Cerebral metabolism following traumatic brain injury: new discoveries with implications for treatment
- PMID: 25709562
- PMCID: PMC4321351
- DOI: 10.3389/fnins.2014.00408
Cerebral metabolism following traumatic brain injury: new discoveries with implications for treatment
Abstract
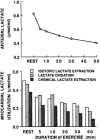
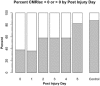
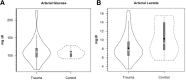
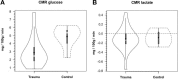
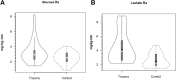
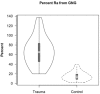
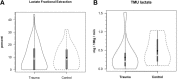
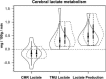
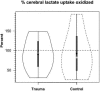
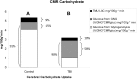
Because it is the product of glycolysis and main substrate for mitochondrial respiration, lactate is the central metabolic intermediate in cerebral energy substrate delivery. Our recent studies on healthy controls and patients following traumatic brain injury (TBI) using [6,6-(2)H2]glucose and [3-(13)C]lactate, along with cerebral blood flow (CBF) and arterial-venous (jugular bulb) difference measurements for oxygen, metabolite levels, isotopic enrichments and (13)CO2 show a massive and previously unrecognized mobilization of lactate from corporeal (muscle, skin, and other) glycogen reserves in TBI patients who were studied 5.7 ± 2.2 days after injury at which time brain oxygen consumption and glucose uptake (CMRO2 and CMRgluc, respectively) were depressed. By tracking the incorporation of the (13)C from lactate tracer we found that gluconeogenesis (GNG) from lactate accounted for 67.1 ± 6.9%, of whole-body glucose appearance rate (Ra) in TBI, which was compared to 15.2 ± 2.8% (mean ± SD, respectively) in healthy, well-nourished controls. Standard of care treatment of TBI patients in state-of-the-art facilities by talented and dedicated heath care professionals reveals presence of a catabolic Body Energy State (BES). Results are interpreted to mean that additional nutritive support is required to fuel the body and brain following TBI. Use of a diagnostic to monitor BES to provide health care professionals with actionable data in providing nutritive formulations to fuel the body and brain and achieve exquisite glycemic control are discussed. In particular, the advantages of using inorganic and organic lactate salts, esters and other compounds are examined. To date, several investigations on brain-injured patients with intact hepatic and renal functions show that compared to dextrose + insulin treatment, exogenous lactate infusion results in normal glycemia.
Keywords: brain fuel; brain metabolism; gluconeogenesis; lactate shuttle; trauma.
Figures

References
-
- Bergman B. C., Butterfield G. E., Wolfel E. E., Lopaschuk G. D., Casazza G. A., Horning M. A., et al. . (1999a). Muscle net glucose uptake and glucose kinetics after endurance training in men. Am. J. Physiol. 277(1 Pt 1): E81–E92. - PubMed
Publication types
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources

