A novel rapid (20-minute) IL-6 release assay using blood mononuclear cells of patients with various clinical forms of drug induced skin injuries
- PMID: 25709746
- PMCID: PMC4326324
- DOI: 10.1186/1939-4551-8-1
A novel rapid (20-minute) IL-6 release assay using blood mononuclear cells of patients with various clinical forms of drug induced skin injuries
Abstract
Background: IL-6 is a pro-inflammatory cytokine which has many well-defined effects. Its synthesis and release from mononuclear cells of drug-sensitized patients was related before to in vitro drug-allergy diagnostics but has not yet been studied in detail.
Methods: The specific release of preformed IL-6 from peripheral blood mononuclear cells (PBMC) after 20 minutes incubation with 0.15-0.5 μM of pure drugs was measured in two groups of drug-allergy suspected donors (159) and respective controls (48). IL-6, TNF-alpha, IL-2, IL-4, IFN-gamma have been measured from cell supernatants by ELISA or by cytometric bead assay. Epicutaneous, intradermal and systemic provocation tests were performed to prove or disprove culprit substances (203 in vivo against 482 in vitro tests). T-test (paired and unpaired); chi2 contingency table; Z statistics and McNemar's test were used to evaluate results.
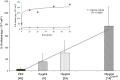
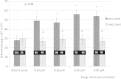
Results: Concanavalin A as positive control released IL-6 from PBMC in linear concentration and exponential time dependent fashion (up to 60 minutes) pointing to the existence of a preformed pool of this cytokine. Preformed IL-6 released at any of 4 standard drug dilutions tested, above 50% over their diluents' levels significantly correlated with the patients' history on drug-induced hypersensitivity symptoms and with in vivo tests. Sensitivity of 85.4% and specificity of 82.4% of the IL-6 release assay were found. The 20' drop in release of TNF-alpha had no diagnostic importance; it has accompanied increased IL-6 release. IL-2, IL-4 and IFN-gamma were undetectable in 20 minutes supernatants. IL-6 release depended on the clinical phenotype but not on the eliciting drug(s) in the molecular mass range of 76-4000 Da. Reactivity of mononuclear cells at the lowest or at multiple drug test concentrations reflected clinical severity per diagnoses and according to area of skin involvement.
Conclusion: This rapid test is applicable to detect a wide scale of drug hypersensitivity.
Keywords: Adverse drug reactions; Drug-induced skin injury; IL-6; Preformed cytokines’ release; T-lymphocytes; TNF-alpha.
Figures




References
-
- De Sousa-Pinto JM, Babo MJ. In vitro diagnosis of drug allergy. Assessment of the Chromatin Activation Test (CAT) Pharmacoepidem Drug Safe. 1997;6(Suppl 3):861–869. - PubMed
LinkOut - more resources
Full Text Sources
Other Literature Sources

