Impact of Enhanced Production of Endogenous Heme Oxygenase-1 by Pitavastatin on Survival and Functional Activities of Bone Marrow-derived Mesenchymal Stem Cells
- PMID: 25714596
- PMCID: PMC4461382
- DOI: 10.1097/FJC.0000000000000231
Impact of Enhanced Production of Endogenous Heme Oxygenase-1 by Pitavastatin on Survival and Functional Activities of Bone Marrow-derived Mesenchymal Stem Cells
Abstract
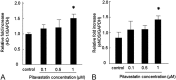
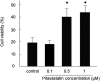
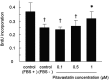
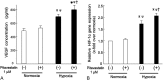
Although mesenchymal stem cells (MSCs) have a therapeutic potential for the repair of tissue injuries, their poor viability in damaged tissue limits their effectiveness. Statins can induce an increased production of heme oxygenase-1 (HO-1), which may prevent this detrimental effect in MSCs. We investigated the protective effect of statin-induced overexpression of HO-1 by examining changes in gene expression and function in MSCs after pitavastatin treatment. The relative expression of the HO-1 and endothelial nitric oxide synthase genes in MSCs was significantly increased after treatment with pitavastatin (MSCs). Immunocytological analysis showed that MSCs also stained with phospho-Akt. After exposure to oxidative stress, MSCs showed increased resistance to induced cell death compared with control MSCs. Under serum starvation conditions, MSCs treated with 1 μM pitavastatin showed enhanced cell proliferation and a marked increase in vascular endothelial growth factor production compared with control MSCs. Interestingly, MSCs showed enhanced tube formation under both normoxia and hypoxia. These results demonstrate that pitavastatin can enhance endogenous HO-1 expression in MSCs, which may protect the cells into the environment of oxidative stress with partial activation of endothelial nitric oxide synthase and Akt phosphorylation.
Conflict of interest statement
Dr. Masakazu Yamagishi received lecture fees from Kowa Pharma Inc. The other authors report no conflicts of interest.
Figures


Similar articles
-
Impact of anti-apoptotic and anti-oxidative effects of bone marrow mesenchymal stem cells with transient overexpression of heme oxygenase-1 on myocardial ischemia.Am J Physiol Heart Circ Physiol. 2010 May;298(5):H1320-9. doi: 10.1152/ajpheart.01330.2008. Epub 2010 Feb 12. Am J Physiol Heart Circ Physiol. 2010. PMID: 20154257
-
Pitavastatin at low dose activates endothelial nitric oxide synthase through PI3K-AKT pathway in endothelial cells.Life Sci. 2005 Mar 25;76(19):2257-68. doi: 10.1016/j.lfs.2004.12.003. Life Sci. 2005. PMID: 15733940
-
Osteogenesis and angiogenesis induced by porous β-CaSiO(3)/PDLGA composite scaffold via activation of AMPK/ERK1/2 and PI3K/Akt pathways.Biomaterials. 2013 Jan;34(1):64-77. doi: 10.1016/j.biomaterials.2012.09.021. Epub 2012 Oct 12. Biomaterials. 2013. PMID: 23069715
-
[Research progress of heme oxygenase 1 in bone tissue engineering].Zhongguo Xiu Fu Chong Jian Wai Ke Za Zhi. 2013 Oct;27(10):1256-61. Zhongguo Xiu Fu Chong Jian Wai Ke Za Zhi. 2013. PMID: 24397142 Review. Chinese.
-
Statins, nitric oxide and neovascularization.Cardiovasc Drug Rev. 2005 Winter;23(4):281-92. doi: 10.1111/j.1527-3466.2005.tb00173.x. Cardiovasc Drug Rev. 2005. PMID: 16614729 Review.
Cited by
-
The Emerging Role of Mesenchymal Stem Cells in Vascular Calcification.Stem Cells Int. 2019 Apr 1;2019:2875189. doi: 10.1155/2019/2875189. eCollection 2019. Stem Cells Int. 2019. PMID: 31065272 Free PMC article. Review.
-
TNF-α promotes survival and migration of MSCs under oxidative stress via NF-κB pathway to attenuate intimal hyperplasia in vein grafts.J Cell Mol Med. 2017 Sep;21(9):2077-2091. doi: 10.1111/jcmm.13131. Epub 2017 Mar 7. J Cell Mol Med. 2017. PMID: 28266177 Free PMC article.
-
Reduced aldehyde dehydrogenase expression in preeclamptic decidual mesenchymal stem/stromal cells is restored by aldehyde dehydrogenase agonists.Sci Rep. 2017 Feb 13;7:42397. doi: 10.1038/srep42397. Sci Rep. 2017. PMID: 28205523 Free PMC article.
-
Overexpression of PRDX2 in Adipose-Derived Mesenchymal Stem Cells Enhances the Therapeutic Effect in a Neurogenic Erectile Dysfunction Rat Model by Inhibiting Ferroptosis.Oxid Med Cell Longev. 2023 Feb 8;2023:4952857. doi: 10.1155/2023/4952857. eCollection 2023. Oxid Med Cell Longev. 2023. PMID: 36819780 Free PMC article.
-
Effects of statins on the biological features of mesenchymal stem cells and therapeutic implications.Heart Fail Rev. 2021 Sep;26(5):1259-1272. doi: 10.1007/s10741-020-09929-9. Heart Fail Rev. 2021. PMID: 32008148 Review.
References
-
- Shibahara S, Yoshizawa M, Suzuki H, et al. Functional analysis of cDNA for two types of human hemeoxygenase and evidence for their separate regulation. J Biochem. 1993;113:214–218. - PubMed
-
- Otterbein LE, Choi AM. Hemeoxygenase: colors of defense against cellular stress. Am J Physiol Lung Cell Mol Physiol. 2000;279:L1029–L1037. - PubMed
-
- Tsubokawa T, Yagi K, Nakanishi C, et al. Impact of anti-apoptotic and ant-oxidative effects of bone marrow mesenchymal stem cells with transient overexpression of heme oxygenase-1 on myocardial ischemia. Am J Physiol Heart Circ Physiol. 2010;298:H1320–H1329. - PubMed
-
- Otterbein LE, Soares MP, Yamashita K, et al. Heme oxygenase-1: unleashing the protective properties of heme. Trends Immunol. 2003;24:449–455. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Medical
Molecular Biology Databases

