Raltegravir non-inferior to nucleoside based regimens in second-line therapy with lopinavir/ritonavir over 96 weeks: a randomised open label study for the treatment of HIV-1 infection
- PMID: 25723472
- PMCID: PMC4344344
- DOI: 10.1371/journal.pone.0118228
Raltegravir non-inferior to nucleoside based regimens in second-line therapy with lopinavir/ritonavir over 96 weeks: a randomised open label study for the treatment of HIV-1 infection
Erratum in
-
Correction: Raltegravir Non-Inferior to Nucleoside Based Regimens in SECOND-LINE Therapy with Lopinavir/Ritonavir over 96 Weeks: A Randomised Open Label Study for the Treatment Of HIV-1 Infection.PLoS One. 2015 Oct 9;10(10):e0140623. doi: 10.1371/journal.pone.0140623. eCollection 2015. PLoS One. 2015. PMID: 26451848 Free PMC article. No abstract available.
Abstract
Objective: To determine the durability over 96 weeks of safety and efficacy of lopinavir/ritonavir (LPV/r) and raltegravir (RAL) which was demonstrated to have non-inferior efficacy relative to a regimen of LPV/r with nucleoside/nucleotide reverse transcriptase inhibitors (N(t)RTIs) (Control) in primary analysis at 48 weeks.
Design: Open label, centrally randomised trial.
Setting: Recruitment was from 37 primary and secondary care sites from Africa, Asia, Australia, Europe and Latin America.
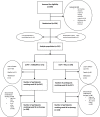
Subjects: 541 HIV-1 infected adults virologically failing first-line non-NRTI + 2N(t)RTI, with no previous exposure to protease inhibitors or integrase strand transfer inhibitors were analysed, 425 completed 96 weeks follow up on randomised therapy.
Intervention: Randomisation was 1:1 to Control or RAL.
Main outcome measures: Differences between the proportion of participants with plasma HIV-1 RNA (VL) <200 copies/mL by intention to treat were compared with a non-inferiority margin of -12%. Differences in biochemical, haematological and metabolic changes were assessed using T-tests.
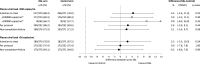
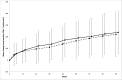
Results: VL <200 copies/mL at 96 weeks was: RAL 80.4%, Control 76.0% (difference: 4.4 [95%CI -2.6, 11.3]) and met non-inferiority criteria. The RAL arm had a significantly higher mean change (difference Control-RAL; 95%CI) in haemoglobin (-2.9; -5.7, -1.1), total lymphocytes (-0.2; -0.3, -0.0), total cholesterol (-0.5; -0.8, -0.3), HDL cholesterol (-0.1; -0.1, -0.0) and LDL cholesterol (-0.3; -0.5, -0.2).
Conclusion: At 96 weeks, both RAL and Control maintained efficacy greater than 75% and continued to demonstrate similar safety profiles. These results support the use of a combination LPV/r and RAL regimen as an option following failure of 1st line NNRTI + 2N(t)RTIs.
Trial registration: ClinicalTrials.gov NCT00931463.
Conflict of interest statement
Figures

References
-
- WHO (2010) Antiretroviral Therapy for HIV Infection in Adults and Adolescents: Recommendations for a Public Health Approach: 2010 Revision. Antiretroviral Therapy for HIV Infection in Adults and Adolescents: Recommendations for a Public Health Approach: 2010 Revision. Geneva.
-
- Boyd MA, Kumarasamy N, Moore CL, Nwizu C, Losso MH, et al. (2013) Ritonavir-boosted lopinavir plus nucleoside or nucleotide reverse transcriptase inhibitors versus ritonavir-boosted lopinavir plus raltegravir for treatment of HIV-1 infection in adults with virological failure of a standard first-line ART regimen (SECOND-LINE): a randomised, open-label, non-inferiority study. Lancet 381: 2091–2099. 10.1016/S0140-6736(13)61164-2 - DOI - PubMed
-
- Levey AS, Bosch JP, Lewis JB, Greene T, Rogers N, et al. (1999) A more accurate method to estimate glomerular filtration rate from serum creatinine: a new prediction equation. Modification of Diet in Renal Disease Study Group. Ann Intern Med 130: 461–470. - PubMed
Publication types
MeSH terms
Substances
Associated data
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Miscellaneous

