Activation of symbiosis signaling by arbuscular mycorrhizal fungi in legumes and rice
- PMID: 25724637
- PMCID: PMC4558648
- DOI: 10.1105/tpc.114.131326
Activation of symbiosis signaling by arbuscular mycorrhizal fungi in legumes and rice
Abstract
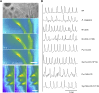
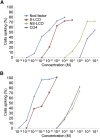
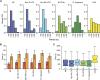
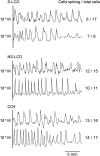
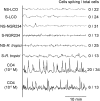
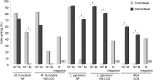
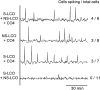
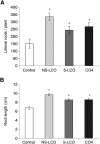
Establishment of arbuscular mycorrhizal interactions involves plant recognition of diffusible signals from the fungus, including lipochitooligosaccharides (LCOs) and chitooligosaccharides (COs). Nitrogen-fixing rhizobial bacteria that associate with leguminous plants also signal to their hosts via LCOs, the so-called Nod factors. Here, we have assessed the induction of symbiotic signaling by the arbuscular mycorrhizal (Myc) fungal-produced LCOs and COs in legumes and rice (Oryza sativa). We show that Myc-LCOs and tetra-acetyl chitotetraose (CO4) activate the common symbiosis signaling pathway, with resultant calcium oscillations in root epidermal cells of Medicago truncatula and Lotus japonicus. The nature of the calcium oscillations is similar for LCOs produced by rhizobial bacteria and by mycorrhizal fungi; however, Myc-LCOs activate distinct gene expression. Calcium oscillations were activated in rice atrichoblasts by CO4, but not the Myc-LCOs, whereas a mix of CO4 and Myc-LCOs activated calcium oscillations in rice trichoblasts. In contrast, stimulation of lateral root emergence occurred following treatment with Myc-LCOs, but not CO4, in M. truncatula, whereas both Myc-LCOs and CO4 were active in rice. Our work indicates that legumes and non-legumes differ in their perception of Myc-LCO and CO signals, suggesting that different plant species respond to different components in the mix of signals produced by arbuscular mycorrhizal fungi.
© 2015 American Society of Plant Biologists. All rights reserved.
Figures


References
-
- Abramoff M.D., Magelhaes P.J., Ram S.J. (2004). Image processing with ImageJ. Biophotonics International 11: 36–42.
-
- Amor B.B., Shaw S.L., Oldroyd G.E., Maillet F., Penmetsa R.V., Cook D., Long S.R., Dénarié J., Gough C. (2003). The NFP locus of Medicago truncatula controls an early step of Nod factor signal transduction upstream of a rapid calcium flux and root hair deformation. Plant J. 34: 495–506. - PubMed
-
- Ané J.M., et al. (2004). Medicago truncatula DMI1 required for bacterial and fungal symbioses in legumes. Science 303: 1364–1367. - PubMed
-
- Banba M., Gutjahr C., Miyao A., Hirochika H., Paszkowski U., Kouchi H., Imaizumi-Anraku H. (2008). Divergence of evolutionary ways among common sym genes: CASTOR and CCaMK show functional conservation between two symbiosis systems and constitute the root of a common signaling pathway. Plant Cell Physiol. 49: 1659–1671. - PubMed
Publication types
MeSH terms
Substances
Associated data
- Actions
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Miscellaneous

