17β-Estradiol increases persistent Na(+) current and excitability of AVPV/PeN Kiss1 neurons in female mice
- PMID: 25734516
- PMCID: PMC4399275
- DOI: 10.1210/me.2014-1392
17β-Estradiol increases persistent Na(+) current and excitability of AVPV/PeN Kiss1 neurons in female mice
Abstract
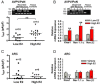
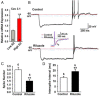
In vitro slice studies have revealed that there are significant differences in the spontaneous firing activity between anteroventral periventricular/periventricular preoptic nucleus (AVPV/PeN) and arcuate nucleus (ARC) kisspeptin (Kiss1) neurons in females. Although both populations express similar endogenous conductances, we have discovered that AVPV/PeN Kiss1 neurons express a subthreshold, persistent sodium current (INaP) that dramatically alters their firing activity. Based on whole-cell recording of Kiss1-Cre-green fluorescent protein (GFP) neurons, INaP was 4-fold greater in AVPV/PeN vs ARC Kiss1 neurons. An LH surge-producing dose of 17β-estradiol (E2) that increased Kiss1 mRNA expression in the AVPV/PeN, also augmented INaP in AVPV/PeN neurons by 2-fold. Because the activation threshold for INaP was close to the resting membrane potential (RMP) of AVPV/PeN Kiss1 neurons (-54 mV), it rendered them much more excitable and spontaneously active vs ARC Kiss1 neurons (RMP = -66 mV). Single-cell RT-PCR revealed that AVPV/PeN Kiss1 neurons expressed the requisite sodium channel α-subunit transcripts, NaV1.1, NaV1.2, and NaV1.6 and β subunits, β2 and β4. Importantly, NaV1.1α and -β2 transcripts in AVPV/PeN, but not ARC, were up-regulated 2- to 3-fold by a surge-producing dose of E2, similar to the transient calcium current channel subunit Cav3.1. The transient calcium current collaborates with INaP to generate burst firing, and selective blockade of INaP by riluzole significantly attenuated rebound burst firing and spontaneous activity. Therefore, INaP appears to play a prominent role in AVPV/PeN Kiss1 neurons to generate spontaneous, repetitive burst firing, which is required for the high-frequency-stimulated release of kisspeptin for exciting GnRH neurons and potentially generating the GnRH surge.
Figures



References
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases

