Repurpose terbutaline sulfate for amyotrophic lateral sclerosis using electronic medical records
- PMID: 25739475
- PMCID: PMC4894399
- DOI: 10.1038/srep08580
Repurpose terbutaline sulfate for amyotrophic lateral sclerosis using electronic medical records
Abstract
Prediction of new disease indications for approved drugs by computational methods has been based largely on the genomics signatures of drugs and diseases. We propose a method for drug repositioning that uses the clinical signatures extracted from over 13 years of electronic medical records from a tertiary hospital, including >9.4 M laboratory tests from >530,000 patients, in addition to diverse genomics signatures. Cross-validation using over 17,000 known drug-disease associations shows this approach outperforms various predictive models based on genomics signatures and a well-known "guilt-by-association" method. Interestingly, the prediction suggests that terbutaline sulfate, which is widely used for asthma, is a promising candidate for amyotrophic lateral sclerosis for which there are few therapeutic options. In vivo tests using zebrafish models found that terbutaline sulfate prevents defects in axons and neuromuscular junction degeneration in a dose-dependent manner. A therapeutic potential of terbutaline sulfate was also observed when axonal and neuromuscular junction degeneration have already occurred in zebrafish model. Cotreatment with a β2-adrenergic receptor antagonist, butoxamine, suggests that the effect of terbutaline is mediated by activation of β2-adrenergic receptors.
Figures
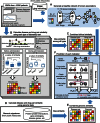


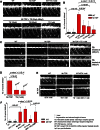
References
-
- Lamb J. et al. The Connectivity Map: using gene-expression signatures to connect small molecules, genes, and disease. Science 313, 1929–1935, 313/5795/1929 [pii]10.1126/science.1132939 (2006). - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Molecular Biology Databases

