A novel role of microglial NADPH oxidase in mediating extra-synaptic function of norepinephrine in regulating brain immune homeostasis
- PMID: 25740080
- PMCID: PMC4405498
- DOI: 10.1002/glia.22801
A novel role of microglial NADPH oxidase in mediating extra-synaptic function of norepinephrine in regulating brain immune homeostasis
Abstract
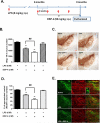
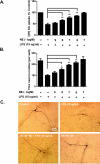
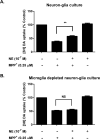
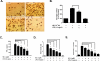
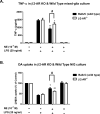
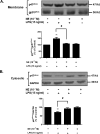
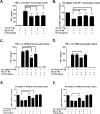
Although the peripheral anti-inflammatory effect of norepinephrine (NE) is well documented, the mechanism by which this neurotransmitter functions as an anti-inflammatory/neuroprotective agent in the central nervous system (CNS) is unclear. This article aimed to determine the anti-inflammatory/neuroprotective effects and underlying mechanisms of NE in inflammation-based dopaminergic neurotoxicity models. In mice, NE-depleting toxin N-(2-chloroethyl)-N-ethyl-2-bromobenzylamine (DSP-4) was injected at 6 months of lipopolysaccharide (LPS)-induced neuroinflammation. It was found that NE depletion enhanced LPS-induced dopaminergic neuron loss in the substantia nigra. This piece of in vivo data prompted us to conduct a series of studies in an effort to elucidate the mechanism as to how NE affects dopamine neuron survival by using primary midbrain neuron/glia cultures. Results showed that submicromolar concentrations of NE dose-dependently protected dopaminergic neurons from LPS-induced neurotoxicity by inhibiting microglia activation and subsequent release of pro-inflammatory factors. However, NE-elicited neuroprotection was not totally abolished in cultures from β2-adrenergic receptor (β2-AR)-deficient mice, suggesting that novel pathways other than β2-AR are involved. To this end, It was found that submicromolar NE dose-dependently inhibited NADPH oxidase (NOX2)-generated superoxide, which contributes to the anti-inflammatory and neuroprotective effects of NE. This novel mechanism was indeed adrenergic receptors independent since both (+) and (-) optic isomers of NE displayed the same potency. We further demonstrated that NE inhibited LPS-induced NOX2 activation by blocking the translocation of its cytosolic subunit to plasma membranes. In summary, we revealed a potential physiological role of NE in maintaining brain immune homeostasis and protecting neurons via a novel mechanism.
Keywords: DSP-4; extra-synaptic; neurodegeneration; neurotransmitter; volume transmission.
© 2015 Wiley Periodicals, Inc.
Figures

Similar articles
-
Clozapine metabolites protect dopaminergic neurons through inhibition of microglial NADPH oxidase.J Neuroinflammation. 2016 May 16;13(1):110. doi: 10.1186/s12974-016-0573-z. J Neuroinflammation. 2016. PMID: 27184631 Free PMC article.
-
Clozapine protects dopaminergic neurons from inflammation-induced damage by inhibiting microglial overactivation.J Neuroimmune Pharmacol. 2012 Mar;7(1):187-201. doi: 10.1007/s11481-011-9309-0. Epub 2011 Aug 26. J Neuroimmune Pharmacol. 2012. PMID: 21870076 Free PMC article.
-
Physiological Concentration of Prostaglandin E2 Exerts Anti-inflammatory Effects by Inhibiting Microglial Production of Superoxide Through a Novel Pathway.Mol Neurobiol. 2018 Oct;55(10):8001-8013. doi: 10.1007/s12035-018-0965-4. Epub 2018 Mar 1. Mol Neurobiol. 2018. PMID: 29492849 Free PMC article.
-
Loss of Brain Norepinephrine Elicits Neuroinflammation-Mediated Oxidative Injury and Selective Caudo-Rostral Neurodegeneration.Mol Neurobiol. 2019 Apr;56(4):2653-2669. doi: 10.1007/s12035-018-1235-1. Epub 2018 Jul 27. Mol Neurobiol. 2019. PMID: 30051353 Free PMC article.
-
Role and mechanism of microglial activation in iron-induced selective and progressive dopaminergic neurodegeneration.Mol Neurobiol. 2014 Jun;49(3):1153-65. doi: 10.1007/s12035-013-8586-4. Epub 2013 Nov 26. Mol Neurobiol. 2014. PMID: 24277523 Free PMC article.
Cited by
-
Dysfunction of the noradrenergic system drives inflammation, α-synucleinopathy, and neuronal loss in mouse colon.Front Immunol. 2023 Feb 10;14:1083513. doi: 10.3389/fimmu.2023.1083513. eCollection 2023. Front Immunol. 2023. PMID: 36845109 Free PMC article.
-
The Association between Glucose 6-Phosphate Dehydrogenase Deficiency and Attention Deficit/Hyperactivity Disorder.Nutrients. 2023 Nov 29;15(23):4948. doi: 10.3390/nu15234948. Nutrients. 2023. PMID: 38068806 Free PMC article.
-
Lipopolysaccharide Effects on Neurotransmission: Understanding Implications for Depression.ACS Chem Neurosci. 2024 Dec 18;15(24):4339-4347. doi: 10.1021/acschemneuro.4c00591. Epub 2024 Nov 27. ACS Chem Neurosci. 2024. PMID: 39601433 Free PMC article. Review.
-
Chemogenetic activation of locus coeruleus neurons ameliorates the severity of multiple sclerosis.J Neuroinflammation. 2023 Sep 1;20(1):198. doi: 10.1186/s12974-023-02865-z. J Neuroinflammation. 2023. PMID: 37658434 Free PMC article.
-
Integrin CD11b mediates locus coeruleus noradrenergic neurodegeneration in a mouse Parkinson's disease model.J Neuroinflammation. 2020 May 6;17(1):148. doi: 10.1186/s12974-020-01823-3. J Neuroinflammation. 2020. PMID: 32375810 Free PMC article.
References
-
- Abercrombie ED, Zigmond MJ. Partial injury to central noradrenergic neurons: reduction of tissue norepinephrine content is greater than reduction of extracellular norepinephrine measured by microdialysis. The Journal of neuroscience : the official journal of the Society for Neuroscience. 1989;9:4062–7. - PMC - PubMed
-
- Antelman SM, Caggiula AR. Briley M, Marien M, editors. Norepinephrine-dopamine interactions: one step beyond. Noradrenergic Mechanisms in Parkinson's Disease. 1993:127–139.
-
- Baloyannis SJ, Costa V, Baloyannis IS. Morphological alterations of the synapses in the locus coeruleus in Parkinson's disease. Journal of the neurological sciences. 2006;248:35–41. - PubMed
-
- Block ML, Hong JS. Microglia and inflammation-mediated neurodegeneration: multiple triggers with a common mechanism. Progress in neurobiology. 2005;76:77–98. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases
Research Materials
Miscellaneous

