Changes in Susceptibility to Oncolytic Vesicular Stomatitis Virus during Progression of Prostate Cancer
- PMID: 25741004
- PMCID: PMC4442527
- DOI: 10.1128/JVI.00257-15
Changes in Susceptibility to Oncolytic Vesicular Stomatitis Virus during Progression of Prostate Cancer
Abstract
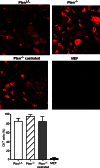
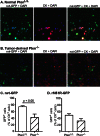
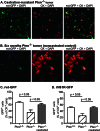
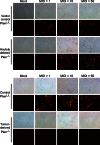
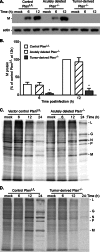
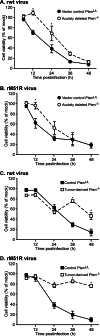
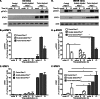
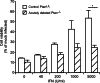
A major challenge to oncolytic virus therapy is that individual cancers vary in their sensitivity to oncolytic viruses, even when these cancers arise from the same tissue type. Variability in response may arise due to differences in the initial genetic lesions leading to cancer development. Alternatively, susceptibility to viral oncolysis may change during cancer progression. These hypotheses were tested using cells from a transgenic mouse model of prostate cancer infected with vesicular stomatitis virus (VSV). Primary cultures from murine cancers derived from prostate-specific Pten deletion contained a mixture of cells that were susceptible and resistant to VSV. Castration-resistant cancers contained a higher percentage of susceptible cells than cancers from noncastrated mice. These results indicate both susceptible and resistant cells can evolve within the same tumor. The role of Pten deletion was further investigated using clonal populations of murine prostate epithelial (MPE) progenitor cells and tumor-derived Pten(-/-) cells. Deletion of Pten in MPE progenitor cells using a lentivirus vector resulted in cells that responded poorly to interferon and were susceptible to VSV infection. In contrast, tumor-derived Pten(-/-) cells expressed higher levels of the antiviral transcription factor STAT1, activated STAT1 in response to VSV, and were resistant to VSV infection. These results suggest that early in tumor development following Pten deletion, cells are primarily sensitive to VSV, but subsequent evolution in tumors leads to development of cells that are resistant to VSV infection. Further evolution in castration-resistant tumors leads to tumors in which cells are primarily sensitive to VSV.
Importance: There has been a great deal of progress in the development of replication-competent viruses that kill cancer cells (oncolytic viruses). However, a major problem is that individual cancers vary in their sensitivity to oncolytic viruses, even when these cancers arise from the same tissue type. The experiments presented here were to determine whether both sensitive and resistant cells are present in prostate cancers originating from a single genetic lesion in transgenic mice, prostate-specific deletion of the gene for the tumor suppressor Pten. The results indicate that murine prostate cancers are composed of both cells that are sensitive and cells that are resistant to oncolytic vesicular stomatitis virus (VSV). Furthermore, androgen deprivation led to castration-resistant prostate cancers that were composed primarily of cells that were sensitive to VSV. These results are encouraging for the use of VSV for the treatment of prostate cancers that are resistant to androgen deprivation therapy.
Copyright © 2015, American Society for Microbiology. All Rights Reserved.
Figures


Similar articles
-
Oncolysis of prostate cancers induced by vesicular stomatitis virus in PTEN knockout mice.Cancer Res. 2010 Feb 15;70(4):1367-76. doi: 10.1158/0008-5472.CAN-09-2377. Epub 2010 Feb 9. Cancer Res. 2010. PMID: 20145134
-
Curcumin promotes the oncoltyic capacity of vesicular stomatitis virus for the treatment of prostate cancers.Virus Res. 2017 Jan 15;228:14-23. doi: 10.1016/j.virusres.2016.10.020. Epub 2016 Nov 16. Virus Res. 2017. PMID: 27865863
-
Interferon Beta and Interferon Alpha 2a Differentially Protect Head and Neck Cancer Cells from Vesicular Stomatitis Virus-Induced Oncolysis.J Virol. 2015 Aug;89(15):7944-54. doi: 10.1128/JVI.00757-15. Epub 2015 May 20. J Virol. 2015. PMID: 25995245 Free PMC article.
-
[Vesicular stomatitis virus in the fight against cancer].Med Sci (Paris). 2013 Feb;29(2):175-82. doi: 10.1051/medsci/2013292015. Epub 2013 Feb 28. Med Sci (Paris). 2013. PMID: 23452604 Review. French.
-
Vesicular stomatitis virus as a flexible platform for oncolytic virotherapy against cancer.J Gen Virol. 2012 Dec;93(Pt 12):2529-2545. doi: 10.1099/vir.0.046672-0. Epub 2012 Oct 10. J Gen Virol. 2012. PMID: 23052398 Free PMC article. Review.
Cited by
-
Recent advances in vesicular stomatitis virus-based oncolytic virotherapy: a 5-year update.J Gen Virol. 2017 Dec;98(12):2895-2911. doi: 10.1099/jgv.0.000980. J Gen Virol. 2017. PMID: 29143726 Free PMC article.
-
Oncogenes: The Passport for Viral Oncolysis Through PKR Inhibition.Biomark Cancer. 2016 Jul 28;8:101-10. doi: 10.4137/BIC.S33378. eCollection 2016. Biomark Cancer. 2016. PMID: 27486347 Free PMC article. Review.
-
Diversity in responses to oncolytic Lassa-vesicular stomatitis virus in patient-derived glioblastoma cells.Mol Ther Oncolytics. 2021 Jun 12;22:232-244. doi: 10.1016/j.omto.2021.06.003. eCollection 2021 Sep 24. Mol Ther Oncolytics. 2021. PMID: 34514102 Free PMC article.
-
PTEN Lipid Phosphatase Activity Enhances Dengue Virus Production through Akt/FoxO1/Maf1 Signaling.Virol Sin. 2021 Jun;36(3):412-423. doi: 10.1007/s12250-020-00291-6. Epub 2020 Oct 12. Virol Sin. 2021. PMID: 33044659 Free PMC article.
-
NEBL and AKT1 maybe new targets to eliminate the colorectal cancer cells resistance to oncolytic effect of vesicular stomatitis virus M-protein.Mol Ther Oncolytics. 2021 Nov 24;23:593-601. doi: 10.1016/j.omto.2021.11.013. eCollection 2021 Dec 17. Mol Ther Oncolytics. 2021. PMID: 34977336 Free PMC article.
References
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical
Research Materials
Miscellaneous

