Neuromedin s-producing neurons act as essential pacemakers in the suprachiasmatic nucleus to couple clock neurons and dictate circadian rhythms
- PMID: 25741729
- PMCID: PMC5811223
- DOI: 10.1016/j.neuron.2015.02.006
Neuromedin s-producing neurons act as essential pacemakers in the suprachiasmatic nucleus to couple clock neurons and dictate circadian rhythms
Abstract
Circadian behavior in mammals is orchestrated by neurons within the suprachiasmatic nucleus (SCN), yet the neuronal population necessary for the generation of timekeeping remains unknown. We show that a subset of SCN neurons expressing the neuropeptide neuromedin S (NMS) plays an essential role in the generation of daily rhythms in behavior. We demonstrate that lengthening period within Nms neurons is sufficient to lengthen period of the SCN and behavioral circadian rhythms. Conversely, mice without a functional molecular clock within Nms neurons lack synchronous molecular oscillations and coherent behavioral daily rhythms. Interestingly, we found that mice lacking Nms and its closely related paralog, Nmu, do not lose in vivo circadian rhythms. However, blocking vesicular transmission from Nms neurons with intact cell-autonomous clocks disrupts the timing mechanisms of the SCN, revealing that Nms neurons define a subpopulation of pacemakers that control SCN network synchrony and in vivo circadian rhythms through intercellular synaptic transmission.
Copyright © 2015 Elsevier Inc. All rights reserved.
Figures

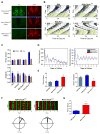

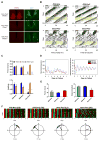
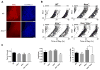



Comment in
-
Short circuiting the circadian system with a new generation of precision tools.Neuron. 2015 Mar 4;85(5):895-8. doi: 10.1016/j.neuron.2015.02.037. Neuron. 2015. PMID: 25741718
Similar articles
-
Expression of the vesicular GABA transporter within neuromedin S+ neurons sustains behavioral circadian rhythms.Proc Natl Acad Sci U S A. 2023 Dec 5;120(49):e2314857120. doi: 10.1073/pnas.2314857120. Epub 2023 Nov 29. Proc Natl Acad Sci U S A. 2023. PMID: 38019855 Free PMC article.
-
Calcium Circadian Rhythmicity in the Suprachiasmatic Nucleus: Cell Autonomy and Network Modulation.eNeuro. 2017 Aug 18;4(4):ENEURO.0160-17.2017. doi: 10.1523/ENEURO.0160-17.2017. eCollection 2017 Jul-Aug. eNeuro. 2017. PMID: 28828400 Free PMC article.
-
Astrocytes Control Circadian Timekeeping in the Suprachiasmatic Nucleus via Glutamatergic Signaling.Neuron. 2017 Mar 22;93(6):1420-1435.e5. doi: 10.1016/j.neuron.2017.02.030. Epub 2017 Mar 9. Neuron. 2017. PMID: 28285822 Free PMC article.
-
Collective timekeeping among cells of the master circadian clock.J Endocrinol. 2016 Jul;230(1):R27-49. doi: 10.1530/JOE-16-0054. Epub 2016 May 6. J Endocrinol. 2016. PMID: 27154335 Free PMC article. Review.
-
Cell autonomy and synchrony of suprachiasmatic nucleus circadian oscillators.Trends Neurosci. 2011 Jul;34(7):349-58. doi: 10.1016/j.tins.2011.05.003. Epub 2011 Jun 12. Trends Neurosci. 2011. PMID: 21665298 Free PMC article. Review.
Cited by
-
Spatiotemporal single-cell analysis of gene expression in the mouse suprachiasmatic nucleus.Nat Neurosci. 2020 Mar;23(3):456-467. doi: 10.1038/s41593-020-0586-x. Epub 2020 Feb 17. Nat Neurosci. 2020. PMID: 32066983
-
GABAergic signalling in the suprachiasmatic nucleus is required for coherent circadian rhythmicity.Eur J Neurosci. 2024 Dec;60(11):6652-6667. doi: 10.1111/ejn.16582. Epub 2024 Nov 18. Eur J Neurosci. 2024. PMID: 39558544 Free PMC article.
-
Interorgan rhythmicity as a feature of healthful metabolism.Cell Metab. 2024 Apr 2;36(4):655-669. doi: 10.1016/j.cmet.2024.01.009. Epub 2024 Feb 8. Cell Metab. 2024. PMID: 38335957 Free PMC article. Review.
-
TRP channels: a missing bond in the entrainment mechanism of peripheral clocks throughout evolution.Temperature (Austin). 2015 Dec 30;2(4):522-34. doi: 10.1080/23328940.2015.1115803. eCollection 2015 Oct-Dec. Temperature (Austin). 2015. PMID: 27227072 Free PMC article. Review.
-
Molecular and Cellular Networks in The Suprachiasmatic Nuclei.Int J Mol Sci. 2019 Apr 25;20(8):2052. doi: 10.3390/ijms20082052. Int J Mol Sci. 2019. PMID: 31027315 Free PMC article. Review.
References
-
- Abrahamson EE, Moore RY. Suprachiasmatic nucleus in the mouse: retinal innervation, intrinsic organization and efferent projections. Brain Res. 2001;916:172–191. - PubMed
-
- Aida R, Moriya T, Araki M, Akiyama M, Wada K, Wada E, Shibata S. Gastrin-releasing peptide mediates photic entrainable signals to dorsal subsets of suprachiasmatic nucleus via induction of Period gene in mice. Mol Pharmacol. 2002;61:26–34. - PubMed
-
- Bae K, Jin X, Maywood ES, Hastings MH, Reppert SM, Weaver DR. Differential functions of mPer1, mPer2, and mPer3 in the SCN circadian clock. Neuron. 2001;30:525–536. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases

