Nuclear sensing of viral DNA, epigenetic regulation of herpes simplex virus infection, and innate immunity
- PMID: 25742715
- PMCID: PMC4424148
- DOI: 10.1016/j.virol.2015.02.009
Nuclear sensing of viral DNA, epigenetic regulation of herpes simplex virus infection, and innate immunity
Abstract
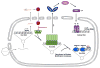
Herpes simplex virus (HSV) undergoes a lytic infection in epithelial cells and a latent infection in neuronal cells, and epigenetic mechanisms play a major role in the differential gene expression under the two conditions. HSV viron DNA is not associated with histones but is rapidly loaded with heterochromatin upon entry into the cell. Viral proteins promote reversal of the epigenetic silencing in epithelial cells while the viral latency-associated transcript promotes additional heterochromatin in neuronal cells. The cellular sensors that initiate the chromatinization of foreign DNA have not been fully defined. IFI16 and cGAS are both essential for innate sensing of HSV DNA, and new evidence shows how they work together to initiate innate signaling. IFI16 also plays a role in the heterochromatinization of HSV DNA, and this review will examine how IFI16 integrates epigenetic regulation and innate sensing of foreign viral DNA to show how these two responses are related.
Keywords: DNA virus; Epigenetics; IFI16; Innate immunity; cGAS.
Copyright © 2015 Elsevier Inc. All rights reserved.
Figures

References
-
- Boutell C, Everett RD. Regulation of alphaherpesvirus infections by the ICP0 family of proteins. J Gen Virol. 2013;94:465–81. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources

