Allyl isothiocyanate induces replication-associated DNA damage response in NSCLC cells and sensitizes to ionizing radiation
- PMID: 25742788
- PMCID: PMC4467145
- DOI: 10.18632/oncotarget.3026
Allyl isothiocyanate induces replication-associated DNA damage response in NSCLC cells and sensitizes to ionizing radiation
Abstract
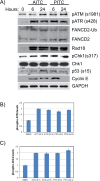
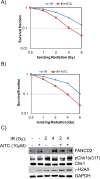
Allyl isothiocyanate (AITC), a constituent of many cruciferous vegetables exhibits significant anticancer activities in many cancer models. Our studies provide novel insights into AITC-induced anticancer mechanisms in human A549 and H1299 non-small cell lung cancer (NSCLC) cells. AITC exposure induced replication stress in NSCLC cells as evidenced by γH2AX and FANCD2 foci, ATM/ATR-mediated checkpoint responses and S and G2/M cell cycle arrest. Furthermore, AITC-induced FANCD2 foci displayed co-localization with BrdU foci, indicating stalled or collapsed replication forks in these cells. Although PITC (phenyl isothiocyanate) exhibited concentration-dependent cytotoxic effects, treatment was less effective compared to AITC. Previously, agents that induce cell cycle arrest in S and G2/M phases were shown to sensitize tumor cells to radiation. Similar to these observations, combination therapy involving AITC followed by radiation treatment exhibited increased DDR and cell killing in NSCLC cells compared to single agent treatment. Combination index (CI) analysis revealed synergistic effects at multiple doses of AITC and radiation, resulting in CI values of less than 0.7 at Fa of 0.5 (50% reduction in survival). Collectively, these studies identify an important anticancer mechanism displayed by AITC, and suggest that the combination of AITC and radiation could be an effective therapy for NSCLC.
Keywords: DNA damage response; dietary isothiocyanates; non-small cell lung cancer; radiosensitization; replication stress.
Figures


Similar articles
-
ERK-modulated intrinsic signaling and G(2)/M phase arrest contribute to the induction of apoptotic death by allyl isothiocyanate in MDA-MB-468 human breast adenocarcinoma cells.Int J Oncol. 2012 Dec;41(6):2065-72. doi: 10.3892/ijo.2012.1640. Epub 2012 Sep 24. Int J Oncol. 2012. PMID: 23008020
-
Cell cycle kinetics, apoptosis rates, DNA damage and TP53 gene expression in bladder cancer cells treated with allyl isothiocyanate (mustard essential oil).Mutat Res. 2014 Apr;762:40-6. doi: 10.1016/j.mrfmmm.2014.02.006. Epub 2014 Mar 10. Mutat Res. 2014. PMID: 24625788
-
Histone deacetylase inhibitors sensitize human non-small cell lung cancer cells to ionizing radiation through acetyl p53-mediated c-myc down-regulation.J Thorac Oncol. 2011 Aug;6(8):1313-9. doi: 10.1097/JTO.0b013e318220caff. J Thorac Oncol. 2011. PMID: 21642861
-
Allyl isothiocyanate regulates oxidative stress, inflammation, cell proliferation, cell cycle arrest, apoptosis, angiogenesis, invasion and metastasis via interaction with multiple cell signaling pathways.Histochem Cell Biol. 2024 Mar;161(3):211-221. doi: 10.1007/s00418-023-02255-9. Epub 2023 Nov 29. Histochem Cell Biol. 2024. PMID: 38019291 Review.
-
Allyl isothiocyanate as a cancer chemopreventive phytochemical.Mol Nutr Food Res. 2010 Jan;54(1):127-35. doi: 10.1002/mnfr.200900323. Mol Nutr Food Res. 2010. PMID: 19960458 Free PMC article. Review.
Cited by
-
Activation of new replication foci under conditions of replication stress.Cell Cycle. 2015;14(16):2634-47. doi: 10.1080/15384101.2015.1064566. Cell Cycle. 2015. PMID: 26212617 Free PMC article.
-
Anticancer effect of selenium/chitosan/polyethylene glycol/allyl isothiocyanate nanocomposites against diethylnitrosamine-induced liver cancer in rats.Saudi J Biol Sci. 2022 May;29(5):3354-3365. doi: 10.1016/j.sjbs.2022.02.012. Epub 2022 Feb 11. Saudi J Biol Sci. 2022. PMID: 35844425 Free PMC article.
-
Eradication of Myrosinase-Tethered Cancer Cells by Allyl Isothiocyanate Derived from Enzymatic Hydrolysis of Sinigrin.Pharmaceutics. 2022 Jan 7;14(1):144. doi: 10.3390/pharmaceutics14010144. Pharmaceutics. 2022. PMID: 35057038 Free PMC article.
-
Allyl Isothiocyanate Exhibits No Anticancer Activity in MDA-MB-231 Breast Cancer Cells.Int J Mol Sci. 2018 Jan 4;19(1):145. doi: 10.3390/ijms19010145. Int J Mol Sci. 2018. PMID: 29300316 Free PMC article.
-
Influence of Cabbage (Brassica oleracea) Accession and Growing Conditions on Myrosinase Activity, Glucosinolates and Their Hydrolysis Products.Foods. 2021 Nov 23;10(12):2903. doi: 10.3390/foods10122903. Foods. 2021. PMID: 34945451 Free PMC article.
References
-
- Siegel R, Naishadham D, Jemal A. Cancer statistics, 2013. CA Cancer J Clin. 2013;63:11–30. - PubMed
-
- Spiro SG, Silvestri GA. One hundred years of lung cancer. Am J Respir Crit Care Med. 2005;172:523–529. - PubMed
-
- Baselga J, Arteaga CL. Critical update and emerging trends in epidermal growth factor receptor targeting in cancer. J Clin Oncol Off J Am Soc Clin Oncol. 2005;23:2445–2459. - PubMed
-
- Bartek J, Mistrik M, Bartkova J. Thresholds of replication stress signaling in cancer development and treatment. Nat Struct Mol Biol. 2012;19:5–7. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials
Miscellaneous

