Real-time measurement of biomagnetic vector fields in functional syncytium using amorphous metal
- PMID: 25744476
- PMCID: PMC4351545
- DOI: 10.1038/srep08837
Real-time measurement of biomagnetic vector fields in functional syncytium using amorphous metal
Abstract
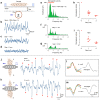
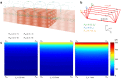
Magnetic field detection of biological electric activities would provide a non-invasive and aseptic estimate of the functional state of cellular organization, namely a syncytium constructed with cell-to-cell electric coupling. In this study, we investigated the properties of biomagnetic waves which occur spontaneously in gut musculature as a typical functional syncytium, by applying an amorphous metal-based gradio-magneto sensor operated at ambient temperature without a magnetic shield. The performance of differentiation was improved by using a single amorphous wire with a pair of transducer coils. Biomagnetic waves of up to several nT were recorded ~1 mm below the sample in a real-time manner. Tetraethyl ammonium (TEA) facilitated magnetic waves reflected electric activity in smooth muscle. The direction of magnetic waves altered depending on the relative angle of the muscle layer and magneto sensor, indicating the existence of propagating intercellular currents. The magnitude of magnetic waves rapidly decreased to ~30% by the initial and subsequent 1 mm separations between sample and sensor. The large distance effect was attributed to the feature of bioelectric circuits constructed by two reverse currents separated by a small distance. This study provides a method for detecting characteristic features of biomagnetic fields arising from a syncytial current.
Conflict of interest statement
The authors declare no competing financial interests.
Figures




References
-
- Szurszewski J. H. Electrical basis for gastrointestinal motility in Physiology of Gastrointestinal Tract 2nd edn. (ed. Johnson, L. R.) 383–422 (Raven, 1987).
-
- Tomita T. Electrical activity (spikes and slow waves) in gastrointestinal smooth muscle in Smooth Muscle: An Assessment of Current Knowledge. (eds. Bülbring, E., Brading, A. F., Jones, A. W. & Tomita, T.) 127–156 (Edward Arnold, 1981).
-
- Wikswo J. P. & Freeman J. A. Magnetic field of a nerve impulse: First measurements. Science 208, 53–55 (1980). - PubMed
-
- Williamson S. J., Lü Z. L., Karron D. & Kaufman L. Advantages and limitations of magnetic source imaging. Brain Topogr. 4, 169–180 (1991). - PubMed
-
- Budker D. & Romalis M. Optical magnetometery. Nat. Phys. 3, 227–234 (2007).
Publication types
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials

