Antiviral effect of mefloquine on feline calicivirus in vitro
- PMID: 25746684
- PMCID: PMC7127475
- DOI: 10.1016/j.vetmic.2015.02.007
Antiviral effect of mefloquine on feline calicivirus in vitro
Abstract
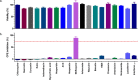
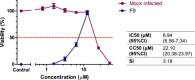
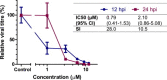
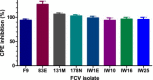
Feline calicivirus (FCV) is an important viral pathogen of domestic cats causing clinical signs ranging from mild to severe oral ulceration or upper respiratory tract disease through to a severe fatal systemic disease. Current therapeutic options are limited, with no direct acting antivirals available for treatment. This study screened a panel of 19 compounds for potential antiviral activity against FCV strain F9 and recent field isolates in vitro. Using a resazurin-based cytopathic effect (CPE) inhibition assay, mefloquine demonstrated a marked inhibitory effect on FCV induced CPE, albeit with a relatively low selectivity index. Orthogonal assays confirmed inhibition of CPE was associated with a significant reduction in viral replication. Mefloquine exhibited a strong inhibitory effect against a panel of seven recent FCV isolates from Australia, with calculated IC50 values for the field isolates approximately 50% lower than against the reference strain FCV F9. In vitro combination therapy with recombinant feline interferon-ω, a biological response modifier currently registered for the treatment of FCV, demonstrated additive effects with a concurrent reduction in the IC50 of mefloquine. These results are the first report of antiviral effects of mefloquine against a calicivirus and support further in vitro and in vivo evaluation of this compound as an antiviral therapeutic for FCV.
Keywords: Antiviral; Cats; Feline calicivirus; Mefloquine.
Copyright © 2015 Elsevier B.V. All rights reserved.
Figures


Similar articles
-
Potential Therapeutic Agents for Feline Calicivirus Infection.Viruses. 2018 Aug 16;10(8):433. doi: 10.3390/v10080433. Viruses. 2018. PMID: 30115859 Free PMC article.
-
Antiviral effect of lithium chloride on feline calicivirus in vitro.Arch Virol. 2015 Dec;160(12):2935-43. doi: 10.1007/s00705-015-2534-8. Epub 2015 Aug 5. Arch Virol. 2015. PMID: 26239340 Free PMC article.
-
In vitro inhibition of field isolates of feline calicivirus with short interfering RNAs (siRNAs).Vet Microbiol. 2015 May 15;177(1-2):78-86. doi: 10.1016/j.vetmic.2015.02.015. Epub 2015 Feb 24. Vet Microbiol. 2015. PMID: 25770890
-
[Treatment of acute viral feline upper respiratory tract infections].Tierarztl Prax Ausg K Kleintiere Heimtiere. 2019 Apr;47(2):98-109. doi: 10.1055/a-0870-0801. Epub 2019 Apr 23. Tierarztl Prax Ausg K Kleintiere Heimtiere. 2019. PMID: 31013527 Review. German.
-
Feline calicivirus.Vet Res. 2007 Mar-Apr;38(2):319-35. doi: 10.1051/vetres:2006056. Epub 2007 Feb 13. Vet Res. 2007. PMID: 17296159 Review.
Cited by
-
Pharmacokinetic Profile of Oral Administration of Mefloquine to Clinically Normal Cats: A Preliminary In-Vivo Study of a Potential Treatment for Feline Infectious Peritonitis (FIP).Animals (Basel). 2020 Jun 8;10(6):1000. doi: 10.3390/ani10061000. Animals (Basel). 2020. PMID: 32521771 Free PMC article.
-
In vitro antiviral effect of germacrone on feline calicivirus.Arch Virol. 2016 Jun;161(6):1559-67. doi: 10.1007/s00705-016-2825-8. Epub 2016 Mar 21. Arch Virol. 2016. PMID: 26997613 Free PMC article.
-
Potential Therapeutic Agents for Feline Calicivirus Infection.Viruses. 2018 Aug 16;10(8):433. doi: 10.3390/v10080433. Viruses. 2018. PMID: 30115859 Free PMC article.
-
Repurposing of FDA Approved Drugs Against SARS-CoV-2 Papain-Like Protease: Computational, Biochemical, and in vitro Studies.Front Microbiol. 2022 May 10;13:877813. doi: 10.3389/fmicb.2022.877813. eCollection 2022. Front Microbiol. 2022. PMID: 35620103 Free PMC article.
-
Progress in the study of mefloquine as an antibiotic adjuvant for combination bacterial inhibition treatment.Front Cell Infect Microbiol. 2024 Nov 28;14:1470891. doi: 10.3389/fcimb.2024.1470891. eCollection 2024. Front Cell Infect Microbiol. 2024. PMID: 39669268 Free PMC article. Review.
References
-
- Feld J.J., Hoofnagle J.H. Mechanism of action of interferon and ribavirin in treatment of hepatitis C. Nature. 2005;436:967–972. - PubMed
-
- Ferraris O., Kessler N., Lina B. Sensitivity of influenza viruses to zanamivir and oseltamivir: a study performed on viruses circulating in France prior to the introduction of neuraminidase inhibitors in clinical practice. Antiviral Res. 2005;68:43–48. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials
Miscellaneous

