Cancer stratification by molecular imaging
- PMID: 25749472
- PMCID: PMC4394457
- DOI: 10.3390/ijms16034918
Cancer stratification by molecular imaging
Abstract
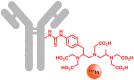
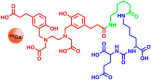
The lack of specificity of traditional cytotoxic drugs has triggered the development of anticancer agents that selectively address specific molecular targets. An intrinsic property of these specialized drugs is their limited applicability for specific patient subgroups. Consequently, the generation of information about tumor characteristics is the key to exploit the potential of these drugs. Currently, cancer stratification relies on three approaches: Gene expression analysis and cancer proteomics, immunohistochemistry and molecular imaging. In order to enable the precise localization of functionally expressed targets, molecular imaging combines highly selective biomarkers and intense signal sources. Thus, cancer stratification and localization are performed simultaneously. Many cancer types are characterized by altered receptor expression, such as somatostatin receptors, folate receptors or Her2 (human epidermal growth factor receptor 2). Similar correlations are also known for a multitude of transporters, such as glucose transporters, amino acid transporters or hNIS (human sodium iodide symporter), as well as cell specific proteins, such as the prostate specific membrane antigen, integrins, and CD20. This review provides a comprehensive description of the methods, targets and agents used in molecular imaging, to outline their application for cancer stratification. Emphasis is placed on radiotracers which are used to identify altered expression patterns of cancer associated markers.
Figures


References
-
- Ummanni R., Mundt F., Pospisil H., Venz S., Scharf C., Barett C., Falth M., Kollermann J., Walther R., Schlomm T., et al. Identification of clinically relevant protein targets in prostate cancer with 2D-DIGE coupled mass spectrometry and systems biology network platform. PLoS One. 2011;6:e16833. doi: 10.1371/journal.pone.0016833. - DOI - PMC - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials
Miscellaneous

