Chronic Deletion and Acute Knockdown of Parkin Have Differential Responses to Acetaminophen-induced Mitophagy and Liver Injury in Mice
- PMID: 25752611
- PMCID: PMC4409255
- DOI: 10.1074/jbc.M114.602284
Chronic Deletion and Acute Knockdown of Parkin Have Differential Responses to Acetaminophen-induced Mitophagy and Liver Injury in Mice
Abstract
We previously demonstrated that pharmacological induction of autophagy protected against acetaminophen (APAP)-induced liver injury in mice by clearing damaged mitochondria. However, the mechanism for removal of mitochondria by autophagy is unknown. Parkin, an E3 ubiquitin ligase, has been shown to be required for mitophagy induction in cultured mammalian cells following mitochondrial depolarization, but its role in vivo is not clear. The purpose of this study was to investigate the role of Parkin-mediated mitophagy in protection against APAP-induced liver injury. We found that Parkin translocated to mitochondria in mouse livers after APAP treatment followed by mitochondrial protein ubiquitination and mitophagy induction. To our surprise, we found that mitophagy still occurred in Parkin knock-out (KO) mice after APAP treatment based on electron microscopy analysis and Western blot analysis for some mitochondrial proteins, and Parkin KO mice were protected against APAP-induced liver injury compared with wild type mice. Mechanistically, we found that Parkin KO mice had decreased activated c-Jun N-terminal kinase (JNK), increased induction of myeloid leukemia cell differentiation protein (Mcl-1) expression, and increased hepatocyte proliferation after APAP treatment in their livers compared with WT mice. In contrast to chronic deletion of Parkin, acute knockdown of Parkin in mouse livers using adenovirus shRNA reduced mitophagy and Mcl-1 expression but increased JNK activation after APAP administration, which exacerbated APAP-induced liver injury. Therefore, chronic deletion (KO) and acute knockdown of Parkin have differential responses to APAP-induced mitophagy and liver injury in mice.
Keywords: Acetaminophen; Autophagy; Cell Death; Liver Injury; Mitochondria; Mitophagy.
© 2015 by The American Society for Biochemistry and Molecular Biology, Inc.
Figures
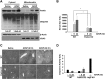

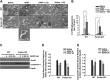



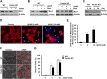

References
-
- Larson A. M. (2007) Acetaminophen hepatotoxicity. Clin. Liver Dis. 11, 525–548 - PubMed
Publication types
MeSH terms
Substances
Grants and funding
- R01 DK102142/DK/NIDDK NIH HHS/United States
- S10 RR027564/RR/NCRR NIH HHS/United States
- P20 GM103549/GM/NIGMS NIH HHS/United States
- P20 GM103418/GM/NIGMS NIH HHS/United States
- 8P20 GM103549/GM/NIGMS NIH HHS/United States
- T32 ES007079/ES/NIEHS NIH HHS/United States
- R01 AA020518/AA/NIAAA NIH HHS/United States
- S10RR027564/RR/NCRR NIH HHS/United States
- 5P20RR021940,/RR/NCRR NIH HHS/United States
- P20 GM104936/GM/NIGMS NIH HHS/United States
- 9P20GM104936/GM/NIGMS NIH HHS/United States
- R37 AA020518/AA/NIAAA NIH HHS/United States
- P20 RR021940/RR/NCRR NIH HHS/United States
LinkOut - more resources
Full Text Sources
Medical
Molecular Biology Databases
Research Materials
Miscellaneous

