Structural characterization of an alternative mode of tigecycline binding to the bacterial ribosome
- PMID: 25753625
- PMCID: PMC4394779
- DOI: 10.1128/AAC.04895-14
Structural characterization of an alternative mode of tigecycline binding to the bacterial ribosome
Abstract
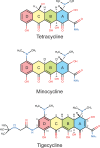
Although both tetracycline and tigecycline inhibit protein synthesis by sterically hindering the binding of tRNA to the ribosomal A site, tigecycline shows increased efficacy in both in vitro and in vivo activity assays and escapes the most common resistance mechanisms associated with the tetracycline class of antibiotics. These differences in activities are attributed to the tert-butyl-glycylamido side chain found in tigecycline. Our structural analysis by X-ray crystallography shows that tigecycline binds the bacterial 30S ribosomal subunit with its tail in an extended conformation and makes extensive interactions with the 16S rRNA nucleotide C1054. These interactions restrict the mobility of C1054 and contribute to the antimicrobial activity of tigecycline, including its resistance to the ribosomal protection proteins.
Copyright © 2015, American Society for Microbiology. All Rights Reserved.
Figures


References
-
- Pioletti M, Schlunzen F, Harms J, Zarivach R, Gluhmann M, Avila H, Bashan A, Bartels H, Auerbach T, Jacobi C, Hartsch T, Yonath A, Franceschi F. 2001. Crystal structures of complexes of the small ribosomal subunit with tetracycline, edeine and IF3. EMBO J 20:1829–1839. doi:10.1093/emboj/20.8.1829. - DOI - PMC - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources

