Neurodegeneration severity can be predicted from early microglia alterations monitored in vivo in a mouse model of chronic glaucoma
- PMID: 25755083
- PMCID: PMC4415894
- DOI: 10.1242/dmm.018788
Neurodegeneration severity can be predicted from early microglia alterations monitored in vivo in a mouse model of chronic glaucoma
Abstract
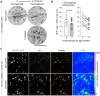
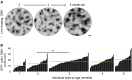
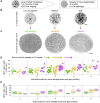
Microglia serve key homeostatic roles, and respond to neuronal perturbation and decline with a high spatiotemporal resolution. The course of all chronic CNS pathologies is thus paralleled by local microgliosis and microglia activation, which begin at early stages of the disease. However, the possibility of using live monitoring of microglia during early disease progression to predict the severity of neurodegeneration has not been explored. Because the retina allows live tracking of fluorescent microglia in their intact niche, here we investigated their early changes in relation to later optic nerve neurodegeneration. To achieve this, we used the DBA/2J mouse model of inherited glaucoma, which develops progressive retinal ganglion cell degeneration of variable severity during aging, and represents a useful model to study pathogenic mechanisms of retinal ganglion cell decline that are similar to those in human glaucoma. We imaged CX3CR1(+/GFP) microglial cells in vivo at ages ranging from 1 to 5 months by confocal scanning laser ophthalmoscopy (cSLO) and quantified cell density and morphological activation. We detected early microgliosis at the optic nerve head (ONH), where axonopathy first manifests, and could track attenuation of this microgliosis induced by minocycline. We also observed heterogeneous and dynamic patterns of early microglia activation in the retina. When the same animals were aged and analyzed for the severity of optic nerve pathology at 10 months of age, we found a strong correlation with the levels of ONH microgliosis at 3 to 4 months. Our findings indicate that live imaging and monitoring the time course and levels of early retinal microgliosis and microglia activation in glaucoma could serve as indicators of future neurodegeneration severity.
Keywords: Confocal ophthalmoscopy; Cx3cr1GFP/+ DBA/2J; Live image analysis; Microglia activation; Microgliosis; Retinal ganglion cells.
© 2015. Published by The Company of Biologists Ltd.
Conflict of interest statement
The authors declare no competing or financial interests.
Figures



References
-
- Alt C., Runnels J. M., Teo G. S. L. and Lin C.P. (2012). In vivo tracking of hematopoietic cells in the retina of chimeric mice with a scanning laser ophthalmoscope. Intravital 1, 132-140 10.4161/intv.23561 - DOI
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Molecular Biology Databases

