Long-lasting complete response to imatinib in a patient with systemic mastocytosis exhibiting wild type KIT
- PMID: 25755909
- PMCID: PMC4351647
Long-lasting complete response to imatinib in a patient with systemic mastocytosis exhibiting wild type KIT
Abstract
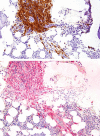
Systemic mastocytosis (SM) is a hematopoietic disorder characterized by abnormal expansion of mast cells (MCs) in visceral organs. The skin is involved in most cases. In adult patients the transforming KIT mutation D816V is usually present and confers resistance against imatinib. Therefore, imatinib is not recommended for patients with KIT D816V+ SM. Nonetheless, imatinib may work in patients with SM lacking KIT D816V. However, little is known about long-term efficacy and safety of this drug in SM. We report on a 62-year-old female patient with indolent SM (ISM) who suffered from severe debilitating skin involvement despite therapy with anti-mediator-type drugs, psoralen and ultraviolet-A-radiation. Although multifocal MC infiltrates were detected in the bone marrow by immunohistochemistry, no KIT mutation was found by sequencing analysis. In 2003, treatment with imatinib (induction, 400 mg/day; maintenance, 200 mg/day) was initiated. During therapy, skin lesions and tryptase levels decreased. Treatment was well tolerated without any side effects. After 10 years, skin lesions have disappeared and the tryptase level is within normal range. This case-study confirms the long-term efficacy and safety of imatinib in patients with SM lacking activating KIT mutations. Imatinib should be considered in select cases of SM in whom MCs exhibit wild-type KIT.
Keywords: KIT D816V; Mastocytosis; drug safety; imatinib; long-term efficacy; tryptase.
Figures

Similar articles
-
Imatinib in systemic mastocytosis: a phase IV clinical trial in patients lacking exon 17 KIT mutations and review of the literature.Oncotarget. 2016 Jul 19;8(40):68950-68963. doi: 10.18632/oncotarget.10711. eCollection 2017 Sep 15. Oncotarget. 2016. PMID: 28978170 Free PMC article.
-
Systemic mastocytosis with KIT V560G mutation presenting as recurrent episodes of vascular collapse: response to disodium cromoglycate and disease outcome.Allergy Asthma Clin Immunol. 2017 Apr 24;13:21. doi: 10.1186/s13223-017-0193-x. eCollection 2017. Allergy Asthma Clin Immunol. 2017. PMID: 28439288 Free PMC article.
-
In vitro and in vivo growth-inhibitory effects of cladribine on neoplastic mast cells exhibiting the imatinib-resistant KIT mutation D816V.Exp Hematol. 2010 Sep;38(9):744-55. doi: 10.1016/j.exphem.2010.05.006. Epub 2010 May 27. Exp Hematol. 2010. PMID: 20553795
-
Novel approaches to treating advanced systemic mastocytosis.Clin Pharmacol. 2019 Jul 10;11:77-92. doi: 10.2147/CPAA.S206615. eCollection 2019. Clin Pharmacol. 2019. PMID: 31372066 Free PMC article. Review.
-
Mastocytosis: state of the art.Pathobiology. 2007;74(2):121-32. doi: 10.1159/000101711. Pathobiology. 2007. PMID: 17587883 Review.
Cited by
-
Advances in the Classification and Treatment of Mastocytosis: Current Status and Outlook toward the Future.Cancer Res. 2017 Mar 15;77(6):1261-1270. doi: 10.1158/0008-5472.CAN-16-2234. Epub 2017 Mar 2. Cancer Res. 2017. PMID: 28254862 Free PMC article. Review.
-
Systemic Mastocytosis: Following the Tyrosine Kinase Inhibition Roadmap.Front Pharmacol. 2020 Apr 14;11:443. doi: 10.3389/fphar.2020.00443. eCollection 2020. Front Pharmacol. 2020. PMID: 32346366 Free PMC article. Review.
-
Imatinib in systemic mastocytosis: a phase IV clinical trial in patients lacking exon 17 KIT mutations and review of the literature.Oncotarget. 2016 Jul 19;8(40):68950-68963. doi: 10.18632/oncotarget.10711. eCollection 2017 Sep 15. Oncotarget. 2016. PMID: 28978170 Free PMC article.
-
Multidisciplinary Challenges in Mastocytosis and How to Address with Personalized Medicine Approaches.Int J Mol Sci. 2019 Jun 18;20(12):2976. doi: 10.3390/ijms20122976. Int J Mol Sci. 2019. PMID: 31216696 Free PMC article. Review.
References
-
- Valent P, Akin C, Sperr WR, Horny HP, Arock M, Lechner K, Bennett JM, Metcalfe DD. Diagnosis and treatment of systemic mastocytosis: state of the art. Br J Haematol. 2003;122:695–717. - PubMed
-
- Akin C, Metcalfe DD. Systemic mastocytosis. Annu Rev Med. 2004;55:419–432. - PubMed
-
- Valent P, Sperr WR, Schwartz LB, Horny HP. Diagnosis and classification of mast cell proliferative disorders: delineation from immunologic diseases and non-mast cell hematopoietic neoplasms. J Allergy Clin Immunol. 2004;114:3–11. - PubMed
-
- Arock M, Valent P. Pathogenesis, classification and treatment of mastocytosis: state of the art in 2010 and future perspectives. Expert Rev Hematol. 2010;3:497–516. - PubMed
-
- Valent P, Horny HP, Escribano L, Longley BJ, Li CY, Schwartz LB, Marone G, Nuñez R, Akin C, Sotlar K, Sperr WR, Wolff K, Brunning RD, Parwaresch RM, Austen KF, Lennert K, Metcalfe DD, Vardiman JW, Bennett JM. Diagnostic criteria and classification of mastocytosis: a consensus proposal. Leuk Res. 2001;25:603–625. - PubMed
LinkOut - more resources
Full Text Sources
