Variation in type of adjuvant chemotherapy received among patients with stage I breast cancer: A multi-institutional study
- PMID: 25757412
- PMCID: PMC4457605
- DOI: 10.1002/cncr.29310
Variation in type of adjuvant chemotherapy received among patients with stage I breast cancer: A multi-institutional study
Abstract
Background: Among patients with stage I breast cancer, there is significant uncertainty concerning the optimal threshold at which to consider chemotherapy, and when considered, there is controversy regarding whether to consider non-intensive versus intensive regimens. The authors examined the types and costs of adjuvant chemotherapy received among patients with stage I breast cancer.
Methods: The current study was a prospective cohort study including patients with stage I breast cancer who were treated at a National Comprehensive Cancer Network center from 2000 through 2009. Stage was defined according to the version of the American Joint Committee on Cancer Staging Manual applicable at the time of diagnosis. Stratifying by human epidermal growth factor receptor 2 (HER2), the authors examined the percentage of patients receiving intensive versus non-intensive chemotherapy regimens and the factors associated with type of chemotherapy administered using multivariable logistic regression. Costs of the most common regimens were estimated.
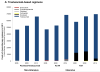
Results: Of 8907 patients, 33% received adjuvant chemotherapy. Among those individuals, there was an increase in the use of intensive chemotherapy within the last decade, from 31% in 2000 through 2005 to 63% in 2008 through 2009 (including an increase in the use of the combination of docetaxel, carboplatin, and trastuzumab) among patients with HER2-positive disease and from 15% in 2000 through 2005 to 41% in 2008 through 2009 among patients with HER2-negative disease (32% of patients with hormone receptor-positive and 59% of patients with triple-negative disease). Among patients treated with non-intensive regimens, there was an increase in the use of the combination of docetaxel and cyclophosphamide noted, with a decrease in the use of the doxorubicin and cyclophosphamide combination. The choice of regimen varied significantly by institution. The major drivers of cost variation were the incorporation of biologics (eg, trastuzumab) and growth factors, with significant variation even within non-intensive and intensive regimens.
Conclusions: Over time, there was an increase in use of intensive regimens among Stage I breast cancer, with striking institutional and cost variations.
Keywords: breast cancer; chemotherapy; cost; institutional variation; stage I.
© 2015 American Cancer Society.
Figures





Similar articles
-
Variation in type of adjuvant chemotherapy received among patients with stage I breast cancer: A multi-institutional Portuguese cohort study.Breast. 2016 Oct;29:68-73. doi: 10.1016/j.breast.2016.07.004. Epub 2016 Jul 25. Breast. 2016. PMID: 27468923
-
Adjuvant trastuzumab with chemotherapy is effective in women with small, node-negative, HER2-positive breast cancer.Cancer. 2011 Dec 15;117(24):5461-8. doi: 10.1002/cncr.26171. Epub 2011 Jun 16. Cancer. 2011. PMID: 21681735
-
Differential response of triple-negative breast cancer to a docetaxel and carboplatin-based neoadjuvant treatment.Cancer. 2010 Sep 15;116(18):4227-37. doi: 10.1002/cncr.25309. Cancer. 2010. PMID: 20549829 Clinical Trial.
-
Neoadjuvant chemotherapy with trastuzumab followed by adjuvant trastuzumab versus neoadjuvant chemotherapy alone, in patients with HER2-positive locally advanced breast cancer (the NOAH trial): a randomised controlled superiority trial with a parallel HER2-negative cohort.Lancet. 2010 Jan 30;375(9712):377-84. doi: 10.1016/S0140-6736(09)61964-4. Lancet. 2010. PMID: 20113825 Clinical Trial.
-
Addition of bevacizumab to three docetaxel regimens as adjuvant therapy for early stage breast cancer.Breast Cancer Res Treat. 2013 Dec;142(3):655-65. doi: 10.1007/s10549-013-2764-y. Epub 2013 Nov 21. Breast Cancer Res Treat. 2013. PMID: 24253810 Clinical Trial.
Cited by
-
Avoiding Peg-Filgrastim Prophylaxis During the Paclitaxel Portion of the Dose-Dense Doxorubicin-Cyclophosphamide and Paclitaxel Regimen: A Prospective Study.J Clin Oncol. 2020 Jul 20;38(21):2390-2397. doi: 10.1200/JCO.19.02484. Epub 2020 Apr 24. J Clin Oncol. 2020. PMID: 32330102 Free PMC article.
-
A nation-wide multicenter 10-year (1999-2008) retrospective study of chemotherapy in Chinese breast cancer patients.Oncotarget. 2017 Mar 22;8(44):75864-75873. doi: 10.18632/oncotarget.16439. eCollection 2017 Sep 29. Oncotarget. 2017. PMID: 29100275 Free PMC article.
References
-
- Chia SK, Speers CH, Bryce CJ, Hayes MM, Olivotto IA. Ten-year outcomes in a population-based cohort of node-negative, lymphatic, and vascular invasion-negative early breast cancers without adjuvant systemic therapies. J Clin Oncol. 2004;22:1630–1637. - PubMed
-
- Rosen PP, Groshen S, Kinne DW, Norton L. Factors influencing prognosis in node-negative breast carcinoma: analysis of 767 T1N0M0/T2N0M0 patients with long-term follow-up. J Clin Oncol. 1993;11:2090–2100. - PubMed
-
- Hanrahan EO, Gonzalez-Angulo AM, Giordano SH, et al. Overall survival and cause-specific mortality of patients with stage T1a,bN0M0 breast carcinoma. J Clin Oncol. 2007;25:4952–4960. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Research Materials
Miscellaneous

