Asexual expansion of Toxoplasma gondii merozoites is distinct from tachyzoites and entails expression of non-overlapping gene families to attach, invade, and replicate within feline enterocytes
- PMID: 25757795
- PMCID: PMC4340605
- DOI: 10.1186/s12864-015-1225-x
Asexual expansion of Toxoplasma gondii merozoites is distinct from tachyzoites and entails expression of non-overlapping gene families to attach, invade, and replicate within feline enterocytes
Abstract
Background: The apicomplexan parasite Toxoplasma gondii is cosmopolitan in nature, largely as a result of its highly flexible life cycle. Felids are its only definitive hosts and a wide range of mammals and birds serve as intermediate hosts. The latent bradyzoite stage is orally infectious in all warm-blooded vertebrates and establishes chronic, transmissible infections. When bradyzoites are ingested by felids, they transform into merozoites in enterocytes and expand asexually as part of their coccidian life cycle. In all other intermediate hosts, however, bradyzoites differentiate exclusively to tachyzoites, and disseminate extraintestinally to many cell types. Both merozoites and tachyzoites undergo rapid asexual population expansion, yet possess different effector fates with respect to the cells and tissues they develop in and the subsequent stages they differentiate into.
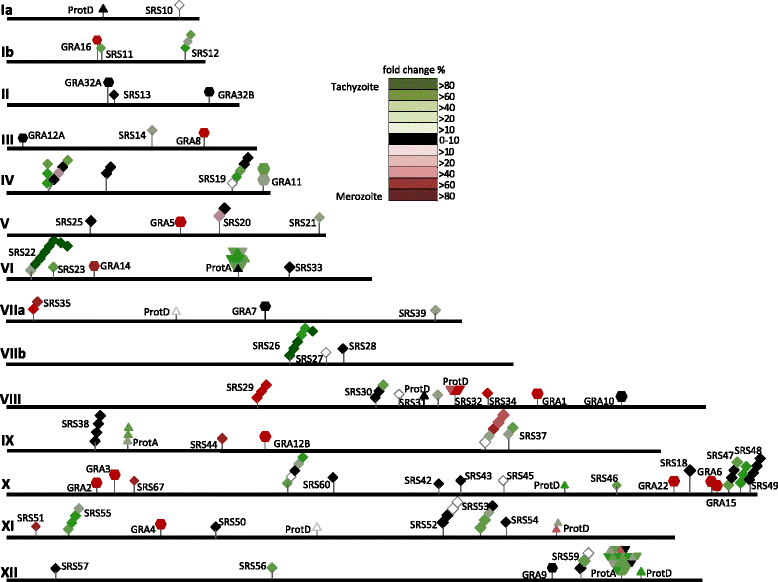
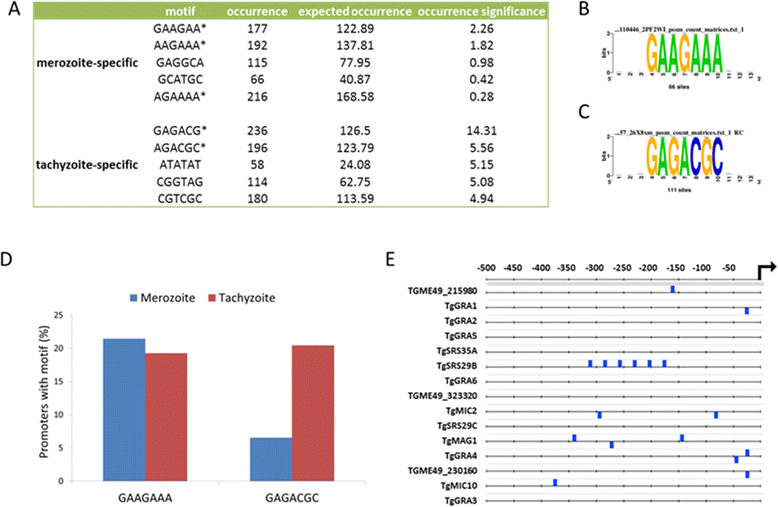
Results: To determine whether merozoites utilize distinct suites of genes to attach, invade, and replicate within feline enterocytes, we performed comparative transcriptional profiling on purified tachyzoites and merozoites. We used high-throughput RNA-Seq to compare the merozoite and tachyzoite transcriptomes. 8323 genes were annotated with sequence reads across the two asexually replicating stages of the parasite life cycle. Metabolism was similar between the two replicating stages. However, significant stage-specific expression differences were measured, with 312 transcripts exclusive to merozoites versus 453 exclusive to tachyzoites. Genes coding for 177 predicted secreted proteins and 64 membrane- associated proteins were annotated as merozoite-specific. The vast majority of known dense-granule (GRA), microneme (MIC), and rhoptry (ROP) genes were not expressed in merozoites. In contrast, a large set of surface proteins (SRS) was expressed exclusively in merozoites.
Conclusions: The distinct expression profiles of merozoites and tachyzoites reveal significant additional complexity within the T. gondii life cycle, demonstrating that merozoites are distinct asexual dividing stages which are uniquely adapted to their niche and biological purpose.
Figures





Similar articles
-
Use of molecular and ultrastructural markers to evaluate stage conversion of Toxoplasma gondii in both the intermediate and definitive host.Int J Parasitol. 2004 Mar 9;34(3):347-60. doi: 10.1016/j.ijpara.2003.11.024. Int J Parasitol. 2004. PMID: 15003495
-
Toxoplasma gondii merozoite gene expression analysis with comparison to the life cycle discloses a unique expression state during enteric development.BMC Genomics. 2014 May 8;15(1):350. doi: 10.1186/1471-2164-15-350. BMC Genomics. 2014. PMID: 24885521 Free PMC article.
-
The merozoite-specific protein, TgGRA11B, identified as a component of the Toxoplasma gondii parasitophorous vacuole in a tachyzoite expression model.Int J Parasitol. 2017 Sep;47(10-11):597-600. doi: 10.1016/j.ijpara.2017.04.001. Epub 2017 May 17. Int J Parasitol. 2017. PMID: 28526607
-
Advances in the life cycle of Toxoplasma gondii.Int J Parasitol. 1998 Jul;28(7):1019-24. doi: 10.1016/s0020-7519(98)00023-x. Int J Parasitol. 1998. PMID: 9724872 Review.
-
[Antigens of Toxoplasma gondii].Wiad Parazytol. 2000;46(3):327-34. Wiad Parazytol. 2000. PMID: 16883687 Review. Polish.
Cited by
-
Transcriptional signatures of clonally derived Toxoplasma tachyzoites reveal novel insights into the expression of a family of surface proteins.PLoS One. 2022 Feb 25;17(2):e0262374. doi: 10.1371/journal.pone.0262374. eCollection 2022. PLoS One. 2022. PMID: 35213559 Free PMC article.
-
Regulation of the developmental programs in Toxoplasma by a novel SNF2L-containing chromatin remodeling complex.Nat Commun. 2025 Jul 1;16(1):5757. doi: 10.1038/s41467-025-60795-1. Nat Commun. 2025. PMID: 40593611 Free PMC article.
-
Secreted protein kinases regulate cyst burden during chronic toxoplasmosis.Cell Microbiol. 2017 Feb;19(2):10.1111/cmi.12651. doi: 10.1111/cmi.12651. Epub 2016 Aug 25. Cell Microbiol. 2017. PMID: 27450947 Free PMC article.
-
In vitro production of cat-restricted Toxoplasma pre-sexual stages.Nature. 2024 Jan;625(7994):366-376. doi: 10.1038/s41586-023-06821-y. Epub 2023 Dec 13. Nature. 2024. PMID: 38093015 Free PMC article.
-
The Structural and Molecular Underpinnings of Gametogenesis in Toxoplasma gondii.Front Cell Infect Microbiol. 2020 Dec 7;10:608291. doi: 10.3389/fcimb.2020.608291. eCollection 2020. Front Cell Infect Microbiol. 2020. PMID: 33365279 Free PMC article. Review.
References
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases

