Emergent properties of composite semiflexible biopolymer networks
- PMID: 25759912
- PMCID: PMC4914020
- DOI: 10.4161/19490992.2014.989035
Emergent properties of composite semiflexible biopolymer networks
Abstract
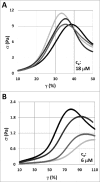
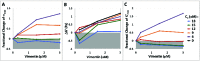
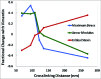
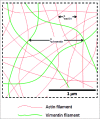
The semiflexible polymers filamentous actin (F-actin) and intermediate filaments (IF) both form complex networks within the cell, and together are key determinants of cellular stiffness. While the mechanics of F-actin networks together with stiff microtubules have been characterized, the interplay between F-actin and IF networks is largely unknown, necessitating the study of composite networks using mixtures of semiflexible biopolymers. We employ bulk rheology in a simplified in vitro system to uncover the fundamental mechanical interactions between networks of the 2 semiflexible polymers, F-actin and vimentin IF. Surprisingly, co-polymerization of actin and vimentin can produce composite networks either stronger or weaker than pure F-actin networks. We show that this effect occurs through steric constraints imposed by IF on F-actin during network formation and filament crosslinking, highlighting novel emergent behavior in composite semiflexible networks.
Keywords: F-actin, filamentous actin; G-actin, globular (monomeric) actin; IF, intermediate filament; actin; composite; intermediate filaments; model systems; networks; rheology; semiflexible polymers; vimentin.
Figures
Similar articles
-
Vimentin intermediate filaments and filamentous actin form unexpected interpenetrating networks that redefine the cell cortex.Proc Natl Acad Sci U S A. 2022 Mar 8;119(10):e2115217119. doi: 10.1073/pnas.2115217119. Epub 2022 Mar 2. Proc Natl Acad Sci U S A. 2022. PMID: 35235449 Free PMC article.
-
Viscoelastic properties of vimentin compared with other filamentous biopolymer networks.J Cell Biol. 1991 Apr;113(1):155-60. doi: 10.1083/jcb.113.1.155. J Cell Biol. 1991. PMID: 2007620 Free PMC article.
-
Dissecting the contribution of actin and vimentin intermediate filaments to mechanical phenotype of suspended cells using high-throughput deformability measurements and computational modeling.J Biomech. 2014 Aug 22;47(11):2598-605. doi: 10.1016/j.jbiomech.2014.05.020. Epub 2014 Jun 6. J Biomech. 2014. PMID: 24952458
-
Polyelectrolyte properties of filamentous biopolymers and their consequences in biological fluids.Soft Matter. 2014 Mar 14;10(10):1439-49. doi: 10.1039/c3sm50854d. Soft Matter. 2014. PMID: 24651463 Free PMC article. Review.
-
Joining actions: crosstalk between intermediate filaments and actin orchestrates cellular physical dynamics and signaling.Sci China Life Sci. 2019 Oct;62(10):1368-1374. doi: 10.1007/s11427-018-9488-1. Epub 2019 May 14. Sci China Life Sci. 2019. PMID: 31098891 Review.
Cited by
-
Thematic Minireview Series: The State of the Cytoskeleton in 2015.J Biol Chem. 2015 Jul 10;290(28):17133-6. doi: 10.1074/jbc.R115.663716. Epub 2015 May 8. J Biol Chem. 2015. PMID: 25957399 Free PMC article. Review.
-
How cytoskeletal crosstalk makes cells move: Bridging cell-free and cell studies.Biophys Rev (Melville). 2024 Jun 3;5(2):021307. doi: 10.1063/5.0198119. eCollection 2024 Jun. Biophys Rev (Melville). 2024. PMID: 38840976 Free PMC article. Review.
-
Intermediate Filaments from Tissue Integrity to Single Molecule Mechanics.Cells. 2021 Jul 27;10(8):1905. doi: 10.3390/cells10081905. Cells. 2021. PMID: 34440673 Free PMC article. Review.
-
Qualitative analysis of contribution of intracellular skeletal changes to cellular elasticity.Cell Mol Life Sci. 2020 Apr;77(7):1345-1355. doi: 10.1007/s00018-019-03328-6. Epub 2019 Oct 11. Cell Mol Life Sci. 2020. PMID: 31605149 Free PMC article. Review.
-
Co-Entangled Actin-Microtubule Composites Exhibit Tunable Stiffness and Power-Law Stress Relaxation.Biophys J. 2018 Sep 18;115(6):1055-1067. doi: 10.1016/j.bpj.2018.08.010. Epub 2018 Aug 16. Biophys J. 2018. PMID: 30177441 Free PMC article.
References
-
- Nogales E. Structural insights into microtubule function. Annu Rev Biochem 2000; 69:277-302; PMID:10966460 - PubMed
-
- Fuchs E, Cleveland DW. A structural scaffolding of intermediate filaments in health and disease. Science 1998; 279:514-9; PMID:9438837, http://dx.doi.org/10.1126/science.279.5350.514 - DOI - PubMed
-
- Fuchs E, Weber K. Intermediate filaments - structure, dynamics, function, and disease. Annu Rev Biochem 1994; 63:345-82; PMID:7979242, http://dx.doi.org/10.1146/annurev.bi.63.070194.002021 - DOI - PubMed
-
- Wang N, Butler JP, Ingber DE. Mechanotransduction across the cell-surface and through the cytoskeleton. Science 1993; 260:1124-7; PMID:7684161, http://dx.doi.org/10.1126/science.7684161 - DOI - PubMed
-
- Smith PG, Deng LH, Fredberg JJ, Maksym GN. Mechanical strain increases cell stiffness through cytoskeletal filament reorganization. Am J Physiol-Lung C 2003; 285:L456-L63; PMID:12704020 - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Miscellaneous
