Action of obestatin in skeletal muscle repair: stem cell expansion, muscle growth, and microenvironment remodeling
- PMID: 25762009
- PMCID: PMC4817756
- DOI: 10.1038/mt.2015.40
Action of obestatin in skeletal muscle repair: stem cell expansion, muscle growth, and microenvironment remodeling
Abstract
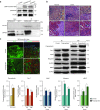
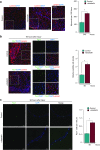
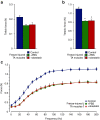
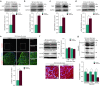
The development of therapeutic strategies for skeletal muscle diseases, such as physical injuries and myopathies, depends on the knowledge of regulatory signals that control the myogenic process. The obestatin/GPR39 system operates as an autocrine signal in the regulation of skeletal myogenesis. Using a mouse model of skeletal muscle regeneration after injury and several cellular strategies, we explored the potential use of obestatin as a therapeutic agent for the treatment of trauma-induced muscle injuries. Our results evidenced that the overexpression of the preproghrelin, and thus obestatin, and GPR39 in skeletal muscle increased regeneration after muscle injury. More importantly, the intramuscular injection of obestatin significantly enhanced muscle regeneration by simulating satellite stem cell expansion as well as myofiber hypertrophy through a kinase hierarchy. Added to the myogenic action, the obestatin administration resulted in an increased expression of vascular endothelial growth factor (VEGF)/vascular endothelial growth factor receptor 2 (VEGFR2) and the consequent microvascularization, with no effect on collagen deposition in skeletal muscle. Furthermore, the potential inhibition of myostatin during obestatin treatment might contribute to its myogenic action improving muscle growth and regeneration. Overall, our data demonstrate successful improvement of muscle regeneration, indicating obestatin is a potential therapeutic agent for skeletal muscle injury and would benefit other myopathies related to muscle regeneration.
Figures





Similar articles
-
Obestatin controls skeletal muscle fiber-type determination.Sci Rep. 2017 May 18;7(1):2137. doi: 10.1038/s41598-017-02337-4. Sci Rep. 2017. PMID: 28522824 Free PMC article.
-
The obestatin/GPR39 system is up-regulated by muscle injury and functions as an autocrine regenerative system.J Biol Chem. 2012 Nov 2;287(45):38379-89. doi: 10.1074/jbc.M112.374926. Epub 2012 Sep 19. J Biol Chem. 2012. PMID: 22992743 Free PMC article.
-
β-Arrestin scaffolds and signaling elements essential for the obestatin/GPR39 system that determine the myogenic program in human myoblast cells.Cell Mol Life Sci. 2016 Feb;73(3):617-35. doi: 10.1007/s00018-015-1994-z. Epub 2015 Jul 27. Cell Mol Life Sci. 2016. PMID: 26211463 Free PMC article.
-
Unlocking the Potential of Obestatin: A Novel Peptide Intervention for Skeletal Muscle Regeneration and Prevention of Atrophy.Mol Biotechnol. 2024 May;66(5):948-959. doi: 10.1007/s12033-023-01011-7. Epub 2024 Jan 10. Mol Biotechnol. 2024. PMID: 38198052 Review.
-
Mechano-biology of skeletal muscle hypertrophy and regeneration: possible mechanism of stretch-induced activation of resident myogenic stem cells.Anim Sci J. 2010 Feb;81(1):11-20. doi: 10.1111/j.1740-0929.2009.00712.x. Anim Sci J. 2010. PMID: 20163667 Review.
Cited by
-
Obestatin controls skeletal muscle fiber-type determination.Sci Rep. 2017 May 18;7(1):2137. doi: 10.1038/s41598-017-02337-4. Sci Rep. 2017. PMID: 28522824 Free PMC article.
-
Potential Therapeutic Effects of Gut Hormones, Ghrelin and Obestatin in Oral Mucositis.Int J Mol Sci. 2019 Mar 27;20(7):1534. doi: 10.3390/ijms20071534. Int J Mol Sci. 2019. PMID: 30934722 Free PMC article. Review.
-
Ghrelin knockout mice display defective skeletal muscle regeneration and impaired satellite cell self-renewal.Endocrine. 2018 Oct;62(1):129-135. doi: 10.1007/s12020-018-1606-4. Epub 2018 May 30. Endocrine. 2018. PMID: 29846901
-
Identification of the Wnt signal peptide that directs secretion on extracellular vesicles.Sci Adv. 2024 Dec 13;10(50):eado5914. doi: 10.1126/sciadv.ado5914. Epub 2024 Dec 11. Sci Adv. 2024. PMID: 39661666 Free PMC article.
-
Development of alginate/chitosan hydrogel loaded with obestatin and evaluation of collagen type I, III, VEGF and TGF-β1 gene expression for skin repair in a rat model (in vitro and in vitro study).Skin Res Technol. 2024 Aug;30(8):e70018. doi: 10.1111/srt.70018. Skin Res Technol. 2024. PMID: 39167033 Free PMC article.
References
-
- Chazaud, B, Brigitte, M, Yacoub-Youssef, H, Arnold, L, Gherardi, R, Sonnet, C et al. (2009). Dual and beneficial roles of macrophages during skeletal muscle regeneration. Exerc Sport Sci Rev 37: 18–22. - PubMed
-
- Tatsumi, R, Anderson, JE, Nevoret, CJ, Halevy, O and Allen, RE (1998). HGF/SF is present in normal adult skeletal muscle and is capable of activating satellite cells. Dev Biol 194: 114–128. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical

