Drug-disease and drug-drug interactions: systematic examination of recommendations in 12 UK national clinical guidelines
- PMID: 25762567
- PMCID: PMC4356453
- DOI: 10.1136/bmj.h949
Drug-disease and drug-drug interactions: systematic examination of recommendations in 12 UK national clinical guidelines
Abstract
Objective: To identify the number of drug-disease and drug-drug interactions for exemplar index conditions within National Institute of Health and Care Excellence (NICE) clinical guidelines.
Design: Systematic identification, quantification, and classification of potentially serious drug-disease and drug-drug interactions for drugs recommended by NICE clinical guidelines for type 2 diabetes, heart failure, and depression in relation to 11 other common conditions and drugs recommended by NICE guidelines for those conditions.
Setting: NICE clinical guidelines for type 2 diabetes, heart failure, and depression
Main outcome measures: Potentially serious drug-disease and drug-drug interactions.
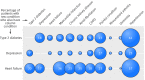
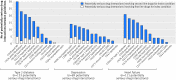
Results: Following recommendations for prescription in 12 national clinical guidelines would result in several potentially serious drug interactions. There were 32 potentially serious drug-disease interactions between drugs recommended in the guideline for type 2 diabetes and the 11 other conditions compared with six for drugs recommended in the guideline for depression and 10 for drugs recommended in the guideline for heart failure. Of these drug-disease interactions, 27 (84%) in the type 2 diabetes guideline and all of those in the two other guidelines were between the recommended drug and chronic kidney disease. More potentially serious drug-drug interactions were identified between drugs recommended by guidelines for each of the three index conditions and drugs recommended by the guidelines for the 11 other conditions: 133 drug-drug interactions for drugs recommended in the type 2 diabetes guideline, 89 for depression, and 111 for heart failure. Few of these drug-disease or drug-drug interactions were highlighted in the guidelines for the three index conditions.
Conclusions: Drug-disease interactions were relatively uncommon with the exception of interactions when a patient also has chronic kidney disease. Guideline developers could consider a more systematic approach regarding the potential for drug-disease interactions, based on epidemiological knowledge of the comorbidities of people with the disease the guideline is focused on, and should particularly consider whether chronic kidney disease is common in the target population. In contrast, potentially serious drug-drug interactions between recommended drugs for different conditions were common. The extensive number of potentially serious interactions requires innovative interactive approaches to the production and dissemination of guidelines to allow clinicians and patients with multimorbidity to make informed decisions about drug selection.
© Dumbreck et al 2015.
Conflict of interest statement
Competing interests: All authors have completed the Unified Competing Interest form at
Figures

Comment in
-
Guidelines, polypharmacy, and drug-drug interactions in patients with multimorbidity.BMJ. 2015 Mar 11;350:h1059. doi: 10.1136/bmj.h1059. BMJ. 2015. PMID: 25761379 No abstract available.
References
-
- Barnett K, Mercer S, Norbury M, Watt G, Wyke S, Guthrie B. Epidemiology of multimorbidity and implications for healthcare, research, and medical education: a cross-sectional study. Lancet 2012;380:37-43 - PubMed
-
- Guthrie B, Payne K, Alderson P, McMurdo M, Mercer S. Adapting clinical guidelines to take account of multimorbidity. BMJ 2012;345:e6341. - PubMed
-
- Guthrie B, McCowan C, Davey P, Simpson CR, Dreischulte T, Barnett K. High risk prescribing in primary care patients particularly vulnerable to adverse drug events: cross sectional population database analysis in Scottish general practice. BMJ 2011;342:d3514. - PubMed
-
- Boyd C, Darer J, Boult C, Fried L, Boult L, Wu A. Clinical practice guidelines and quality of care for older patients with multiple comorbid diseases implications for pay for performance. JAMA 2005;294:716-24 - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
