Impact of eNOS-Dependent Oxidative Stress on Endothelial Function and Neointima Formation
- PMID: 25764009
- PMCID: PMC4580305
- DOI: 10.1089/ars.2014.6059
Impact of eNOS-Dependent Oxidative Stress on Endothelial Function and Neointima Formation
Abstract
Aims: Vascular oxidative stress generated by endothelial NO synthase (eNOS) was observed in experimental and clinical cardiovascular disease, but its relative importance for vascular pathologies is unclear. We investigated the impact of eNOS-dependent vascular oxidative stress on endothelial function and on neointimal hyperplasia.
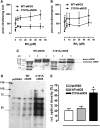
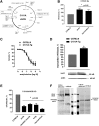
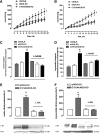
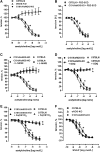
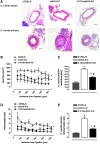
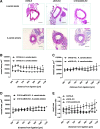
Results: A dimer-destabilized mutant of bovine eNOS where cysteine 101 was replaced by alanine was cloned and introduced into an eNOS-deficient mouse strain (eNOS-KO) in an endothelial-specific manner. Destabilization of mutant eNOS in cells and eNOS-KO was confirmed by the reduced dimer/monomer ratio. Purified mutant eNOS and transfected cells generated less citrulline and NO, respectively, while superoxide generation was enhanced. In eNOS-KO, introduction of mutant eNOS caused a 2.3-3.7-fold increase in superoxide and peroxynitrite formation in the aorta and myocardium. This was completely blunted by an NOS inhibitor. Nevertheless, expression of mutant eNOS in eNOS-KO completely restored maximal aortic endothelium-dependent relaxation to acetylcholine. Neointimal hyperplasia induced by carotid binding was much larger in eNOS-KO than in mutant eNOS-KO and C57BL/6, while the latter strains showed comparable hyperplasia. Likewise, vascular remodeling was blunted in eNOS-KO only.
Innovation: Our results provide the first in vivo evidence that eNOS-dependent oxidative stress is unlikely to be an initial cause of impaired endothelium-dependent vasodilation and/or a pathologic factor promoting intimal hyperplasia. These findings highlight the importance of other sources of vascular oxidative stress in cardiovascular disease.
Conclusion: eNOS-dependent oxidative stress is unlikely to induce functional vascular damage as long as concomitant generation of NO is preserved. This underlines the importance of current and new therapeutic strategies in improving endothelial NO generation.
Figures
Similar articles
-
Selective impairment of blood pressure reduction by endothelial nitric oxide synthase dimer destabilization in mice.J Hypertens. 2017 Jan;35(1):76-88. doi: 10.1097/HJH.0000000000001127. J Hypertens. 2017. PMID: 27861245
-
Chronic cigarette smoke exposure triggers a vicious cycle of leukocyte and endothelial-mediated oxidant stress that results in vascular dysfunction.Am J Physiol Heart Circ Physiol. 2020 Jul 1;319(1):H51-H65. doi: 10.1152/ajpheart.00657.2019. Epub 2020 May 15. Am J Physiol Heart Circ Physiol. 2020. PMID: 32412791 Free PMC article.
-
Red blood cells from endothelial nitric oxide synthase-deficient mice induce vascular dysfunction involving oxidative stress and endothelial arginase I.Redox Biol. 2023 Apr;60:102612. doi: 10.1016/j.redox.2023.102612. Epub 2023 Jan 13. Redox Biol. 2023. PMID: 36681048 Free PMC article.
-
Sustained hypertension despite endothelial-specific eNOS rescue in eNOS-deficient mice.Biochem Biophys Res Commun. 2015 Mar 13;458(3):576-583. doi: 10.1016/j.bbrc.2015.01.152. Epub 2015 Feb 11. Biochem Biophys Res Commun. 2015. PMID: 25680465
-
Caloric restriction reverses high-fat diet-induced endothelial dysfunction and vascular superoxide production in C57Bl/6 mice.Heart Vessels. 2010 May;25(3):254-62. doi: 10.1007/s00380-009-1182-x. Epub 2010 May 29. Heart Vessels. 2010. PMID: 20512454
Cited by
-
Intralesional Infiltrations of Arteriosclerotic Tissue Cells-Free Filtrate Reproduce Vascular Pathology in Healthy Recipient Rats.Int J Mol Sci. 2022 Jan 28;23(3):1511. doi: 10.3390/ijms23031511. Int J Mol Sci. 2022. PMID: 35163435 Free PMC article.
-
Acute effects of cigarette smoke on Endothelial Nitric Oxide synthase, vascular cell adhesion molecule 1 and aortic intima media thickness.F1000Res. 2023 Oct 10;10:396. doi: 10.12688/f1000research.28375.4. eCollection 2021. F1000Res. 2023. PMID: 38046985 Free PMC article.
-
Oxidative stress and its role in recurrent pregnancy loss: mechanisms and implications.J Mol Histol. 2024 Dec 26;56(1):55. doi: 10.1007/s10735-024-10332-z. J Mol Histol. 2024. PMID: 39724438 Review.
-
Molecular and Biochemical Mechanisms of Cardiomyopathy Development Following Prenatal Hypoxia-Focus on the NO System.Antioxidants (Basel). 2025 Jun 16;14(6):743. doi: 10.3390/antiox14060743. Antioxidants (Basel). 2025. PMID: 40563375 Free PMC article. Review.
-
Possibility of Using NO Modulators for Pharmacocorrection of Endothelial Dysfunction After Prenatal Hypoxia.Pharmaceuticals (Basel). 2025 Jan 16;18(1):106. doi: 10.3390/ph18010106. Pharmaceuticals (Basel). 2025. PMID: 39861168 Free PMC article.
References
-
- This reference has been deleted.
-
- Boger RH. Asymmetric dimethylarginine (ADMA): a novel risk marker in cardiovascular medicine and beyond. Ann Med 38: 126–136, 2006 - PubMed
-
- Bonaa KH, Njolstad I, Ueland PM, Schirmer H, Tverdal A, Steigen T, Wang H, Nordrehaug JE, Arnesen E, and Rasmussen K. Homocysteine lowering and cardiovascular events after acute myocardial infarction. N Engl J Med 354: 1578–1588, 2006 - PubMed
-
- Chen PF, Tsai AL, and Wu KK. Cysteine 99 of endothelial nitric oxide synthase (NOS-III) is critical for tetrahydrobiopterin-dependent NOS-III stability and activity. Biochem Biophys Res Commun 215: 1119–1129, 1995 - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials

