The endogenous cell-fate factor dachshund restrains prostate epithelial cell migration via repression of cytokine secretion via a cxcl signaling module
- PMID: 25769723
- PMCID: PMC4433595
- DOI: 10.1158/0008-5472.CAN-14-0611
The endogenous cell-fate factor dachshund restrains prostate epithelial cell migration via repression of cytokine secretion via a cxcl signaling module
Abstract
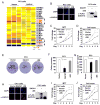
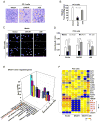
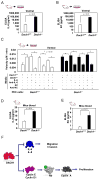
Prostate cancer is the second leading form of cancer-related death in men. In a subset of prostate cancer patients, increased chemokine signaling IL8 and IL6 correlates with castrate-resistant prostate cancer (CRPC). IL8 and IL6 are produced by prostate epithelial cells and promote prostate cancer cell invasion; however, the mechanisms restraining prostate epithelial cell cytokine secretion are poorly understood. Herein, the cell-fate determinant factor DACH1 inhibited CRPC tumor growth in mice. Using Dach1(fl/fl)/Probasin-Cre bitransgenic mice, we show IL8 and IL6 secretion was altered by approximately 1,000-fold by endogenous Dach1. Endogenous Dach1 is shown to serve as a key endogenous restraint to prostate epithelial cell growth and restrains migration via CXCL signaling. DACH1 inhibited expression, transcription, and secretion of the CXCL genes (IL8 and IL6) by binding to their promoter regulatory regions in chromatin. DACH1 is thus a newly defined determinant of benign and malignant prostate epithelium cellular growth, migration, and cytokine abundance in vivo.
©2015 American Association for Cancer Research.
Conflict of interest statement
Conflicts of Interest: R.G.P. holds major (> $10,000) ownership interests in, and serves as CSO/Founder of the biopharmaceutical companies ProstaGene, LLC and AAA Phoenix, Inc. R.G.P. additionally holds ownership interests (value unknown) for several granted and submitted patent applications.
Figures


Similar articles
-
DACH1 is a novel predictive and prognostic biomarker in hepatocellular carcinoma as a negative regulator of Wnt/β-catenin signaling.Oncotarget. 2015 Apr 20;6(11):8621-34. doi: 10.18632/oncotarget.3281. Oncotarget. 2015. PMID: 25940701 Free PMC article.
-
DACH1 is a cell fate determination factor that inhibits cyclin D1 and breast tumor growth.Mol Cell Biol. 2006 Oct;26(19):7116-29. doi: 10.1128/MCB.00268-06. Mol Cell Biol. 2006. PMID: 16980615 Free PMC article.
-
Dachshund inhibits oncogene-induced breast cancer cellular migration and invasion through suppression of interleukin-8.Proc Natl Acad Sci U S A. 2008 May 13;105(19):6924-9. doi: 10.1073/pnas.0802085105. Epub 2008 May 8. Proc Natl Acad Sci U S A. 2008. PMID: 18467491 Free PMC article.
-
Suppression of DACH1 promotes migration and invasion of colorectal cancer via activating TGF-β-mediated epithelial-mesenchymal transition.Biochem Biophys Res Commun. 2015 May 1;460(2):314-9. doi: 10.1016/j.bbrc.2015.03.032. Epub 2015 Mar 14. Biochem Biophys Res Commun. 2015. PMID: 25778865
-
Multiple roles of chemokine (C-C motif) ligand 2 in promoting prostate cancer growth.J Natl Cancer Inst. 2010 Apr 21;102(8):522-8. doi: 10.1093/jnci/djq044. Epub 2010 Mar 16. J Natl Cancer Inst. 2010. PMID: 20233997 Free PMC article. Review.
Cited by
-
Dach1 transcription factor regulates the expression of peripheral node addressin and lymphocyte trafficking in lymph nodes.Curr Res Immunol. 2022 Aug 21;3:175-185. doi: 10.1016/j.crimmu.2022.08.008. eCollection 2022. Curr Res Immunol. 2022. PMID: 36045707 Free PMC article.
-
The retinal determination gene network: from developmental regulator to cancer therapeutic target.Oncotarget. 2016 Aug 2;7(31):50755-50765. doi: 10.18632/oncotarget.9394. Oncotarget. 2016. PMID: 27203207 Free PMC article. Review.
-
Transcriptome-wide association analysis identifies DACH1 as a kidney disease risk gene that contributes to fibrosis.J Clin Invest. 2021 May 17;131(10):e141801. doi: 10.1172/JCI141801. J Clin Invest. 2021. PMID: 33998598 Free PMC article.
-
Retinal determination gene networks: from biological functions to therapeutic strategies.Biomark Res. 2023 Feb 8;11(1):18. doi: 10.1186/s40364-023-00459-8. Biomark Res. 2023. PMID: 36750914 Free PMC article. Review.
-
DACH1 antagonizes CXCL8 to repress tumorigenesis of lung adenocarcinoma and improve prognosis.J Hematol Oncol. 2018 Apr 10;11(1):53. doi: 10.1186/s13045-018-0597-1. J Hematol Oncol. 2018. PMID: 29636079 Free PMC article.
References
-
- Jemal A, Siegel R, Xu J, Ward E. 2010 Cancer statistics. CA Cancer J Clin. 2010;60:277–300. - PubMed
-
- Eisenberger MA. The Current Knowledge of Hormonal Therapy in the Treatment of Prostate Cancer. In: Pestell RG, Nevalainen MT, editors. Prostate Cancer - Signaling Networks, Genetics, and New Treatment Strategies. Santa Monica, CA: Humana Press; 2008. pp. 339–354.
-
- Heinlein CA, Chang C. Androgen receptor in prostate cancer. Endocr Rev. 2004;25:276–308. - PubMed
-
- Miyamoto H, Messing EM, Chang C. Does androgen deprivation improve treatment outcomes in patients with low-risk and intermediate-risk prostate cancer? Nat Clin Pract Oncol. 2005;2:236–237. - PubMed
-
- Chang CS, Kokontis J, Liao ST. Molecular cloning of human and rat complementary DNA encoding androgen receptors. Science. 1998;240:324–326. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Molecular Biology Databases

