Carbon monoxide negatively regulates NLRP3 inflammasome activation in macrophages
- PMID: 25770182
- PMCID: PMC4437010
- DOI: 10.1152/ajplung.00400.2014
Carbon monoxide negatively regulates NLRP3 inflammasome activation in macrophages
Abstract
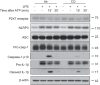
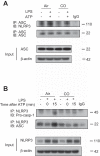
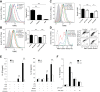
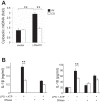
Inflammasomes are cytosolic protein complexes that promote the cleavage of caspase-1, which leads to the maturation and secretion of proinflammatory cytokines, including interleukin-1β (IL-1β) and IL-18. Among the known inflammasomes, the nucleotide-binding domain, leucine-rich-containing family, pyrin domain-containing-3 (NLRP3)-dependent inflammasome is critically involved in the pathogenesis of various acute or chronic inflammatory diseases. Carbon monoxide (CO), a gaseous molecule physiologically produced in cells and tissues during heme catabolism, can act as an anti-inflammatory molecule and a potent negative regulator of Toll-like receptor signaling pathways. To date, the role of CO in inflammasome-mediated immune responses has not been fully investigated. Here, we demonstrated that CO inhibited caspase-1 activation and the secretion of IL-1β and IL-18 in response to lipopolysaccharide (LPS) and ATP treatment in bone marrow-derived macrophages. CO also inhibited IL-18 secretion in response to LPS and nigericin treatment, another NLRP3 inflammasome activation model. In contrast, CO did not suppress IL-18 secretion in response to LPS and poly(dA:dT), an absent in melanoma 2 (AIM2)-mediated inflammasome model. LPS and ATP stimulation induced the formation of complexes between NLRP3 and apoptosis-associated speck-like protein, or NLRP3 and caspase-1. CO treatment inhibited these molecular interactions that were induced by LPS and ATP. Furthermore, CO inhibited mitochondrial ROS generation and the decrease of mitochondrial membrane potential induced by LPS and ATP in macrophages. We also observed that the inhibitory effect of CO on the translocation of mitochondrial DNA into the cytosol was associated with suppression of cytokine secretion. Our results suggest that CO negatively regulates NLRP3 inflammasome activation by preventing mitochondrial dysfunction.
Keywords: interleukin-18; mitochondria; nucleotide-binding domain, leucine-rich-containing family, pyrin domain-containing-3.
Copyright © 2015 the American Physiological Society.
Figures


References
-
- Abramson SB, Amin A. Blocking the effects of IL-1 in rheumatoid arthritis protects bone and cartilage. Rheumatology (Oxford) 41: 972–980, 2002. - PubMed
-
- Bilban M, Bach FH, Otterbein SL, Ifedigbo E, d'Avila JC, Esterbauer H, Chin BY, Usheva A, Robson SC, Wagner O, Otterbein LE. Carbon monoxide orchestrates a protective response through PPARgamma. Immunity 24: 601–610, 2006. - PubMed
-
- Bindu S, Pal C, Dey S, Goyal M, Alam A, Iqbal MS, Dutta S, Sarkar S, Kumar R, Maity P, Bandyopadhyay U. Translocation of heme oxygenase-1 to mitochondria is a novel cytoprotective mechanism against non-steroidal anti-inflammatory drug-induced mitochondrial oxidative stress, apoptosis, and gastric mucosal injury. J Biol Chem 286: 39387–39402, 2011. - PMC - PubMed
-
- Boraschi D, Dinarello CA. IL18 in autoimmunity: review. Eur Cytokine Network 17: 224–252, 2006. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Miscellaneous

