Determination of base binding strength and base stacking interaction of DNA duplex using atomic force microscope
- PMID: 25772017
- PMCID: PMC4360479
- DOI: 10.1038/srep09143
Determination of base binding strength and base stacking interaction of DNA duplex using atomic force microscope
Abstract
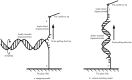
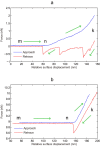
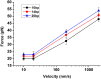
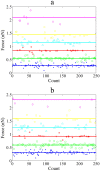
As one of the most crucial properties of DNA, the structural stability and the mechanical strength are attracting a great attention. Here, we take advantage of high force resolution and high special resolution of Atom Force Microscope and investigate the mechanical force of DNA duplexes. To evaluate the base pair hydrogen bond strength and base stacking force in DNA strands, we designed two modes (unzipping and stretching) for the measurement rupture forces. Employing k-means clustering algorithm, the ruptured force are clustered and the mean values are estimated. We assessed the influence of experimental parameters and performed the force evaluation for DNA duplexes of pure dG/dC and dA/dT base pairs. The base binding strength of single dG/dC and single dA/dT were estimated to be 20.0 ± 0.2 pN and 14.0 ± 0.3 pN, respectively, and the base stacking interaction was estimated to be 2.0 ± 0.1 pN. Our results provide valuable information about the quantitative evaluation of the mechanical properties of the DNA duplexes.
Figures
Similar articles
-
Sequence-dependent mechanics of single DNA molecules.Nat Struct Biol. 1999 Apr;6(4):346-9. doi: 10.1038/7582. Nat Struct Biol. 1999. PMID: 10201403
-
Dynamics of DNA duplexes containing internal G.T, G.A, A.C, and T.C pairs: hydrogen exchange at and adjacent to mismatch sites.Fed Proc. 1984 Aug;43(11):2663-70. Fed Proc. 1984. PMID: 6745451
-
DNA base pair resolution by single molecule force spectroscopy.Nucleic Acids Res. 2004 Sep 14;32(16):4876-83. doi: 10.1093/nar/gkh826. Print 2004. Nucleic Acids Res. 2004. PMID: 15367697 Free PMC article.
-
Denaturation transition of stretched DNA.Biochem Soc Trans. 2013 Apr;41(2):639-45. doi: 10.1042/BST20120298. Biochem Soc Trans. 2013. PMID: 23514169 Review.
-
An appreciation of John D. Ferry and the mechanical properties of single molecules.Biophys Chem. 2004 Dec 20;112(2-3):105-8. doi: 10.1016/j.bpc.2004.07.044. Biophys Chem. 2004. PMID: 15688527 Review. No abstract available.
Cited by
-
Coarse-Grained Simulations of DNA Reveal Angular Dependence of Sticky-End Binding.J Phys Chem B. 2021 Apr 29;125(16):4016-4024. doi: 10.1021/acs.jpcb.1c00432. Epub 2021 Apr 19. J Phys Chem B. 2021. PMID: 33870695 Free PMC article.
-
Molecular Recognition in Confined Space Elucidated with DNA Nanopores and Single-Molecule Force Microscopy.Nano Lett. 2023 May 24;23(10):4439-4447. doi: 10.1021/acs.nanolett.3c00743. Epub 2023 May 11. Nano Lett. 2023. PMID: 37166380 Free PMC article.
-
Hydrogen Bonding in Natural and Unnatural Base Pairs-A Local Vibrational Mode Study.Molecules. 2021 Apr 14;26(8):2268. doi: 10.3390/molecules26082268. Molecules. 2021. PMID: 33919989 Free PMC article.
-
Label-Free Oligonucleotide-Based SPR Biosensor for the Detection of the Gene Mutation Causing Prothrombin-Related Thrombophilia.Sensors (Basel). 2020 Oct 31;20(21):6240. doi: 10.3390/s20216240. Sensors (Basel). 2020. PMID: 33142935 Free PMC article.
-
Hydrogel-based molecular tension fluorescence microscopy for investigating receptor-mediated rigidity sensing.Nat Methods. 2023 Nov;20(11):1780-1789. doi: 10.1038/s41592-023-02037-0. Epub 2023 Oct 5. Nat Methods. 2023. PMID: 37798478
References
-
- Smith S. B., Finzi L. & Bustamante C. Direct mechanical measurements of the elasticity of single DNA molecules by using magnetic beads. Science 258, 1122–6 (1992). - PubMed
-
- Rudi K. et al. Rapid, universal method to isolate PCR-ready DNA using magnetic beads. Biotechniques 22, 506–11 (1997). - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials

