Na+ channel function, regulation, structure, trafficking and sequestration
- PMID: 25772290
- PMCID: PMC4376415
- DOI: 10.1113/jphysiol.2014.281428
Na+ channel function, regulation, structure, trafficking and sequestration
Abstract
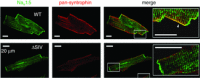
This paper is the second of a series of three reviews published in this issue resulting from the University of California Davis Cardiovascular Symposium 2014: Systems approach to understanding cardiac excitation-contraction coupling and arrhythmias: Na(+) channel and Na(+) transport. The goal of the symposium was to bring together experts in the field to discuss points of consensus and controversy on the topic of sodium in the heart. The present review focuses on Na(+) channel function and regulation, Na(+) channel structure and function, and Na(+) channel trafficking, sequestration and complexing.
© 2014 The Authors. The Journal of Physiology © 2014 The Physiological Society.
Figures

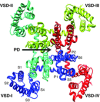
References
-
- Bahler M. Rhoads A. Calmodulin signaling via the IQ motif. FEBS Lett. 2002;513:107–113. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources

