Dynamics of Sun5 localization during spermatogenesis in wild type and Dpy19l2 knock-out mice indicates that Sun5 is not involved in acrosome attachment to the nuclear envelope
- PMID: 25775128
- PMCID: PMC4361733
- DOI: 10.1371/journal.pone.0118698
Dynamics of Sun5 localization during spermatogenesis in wild type and Dpy19l2 knock-out mice indicates that Sun5 is not involved in acrosome attachment to the nuclear envelope
Erratum in
-
Correction: Dynamics of Sun5 localization during spermatogenesis in wild type and Dpy19l2 knock-out mice indicates that Sun5 is not involved in acrosome attachment to the nuclear envelope.PLoS One. 2015 Apr 10;10(4):e0125452. doi: 10.1371/journal.pone.0125452. eCollection 2015. PLoS One. 2015. PMID: 25860994 Free PMC article. No abstract available.
Abstract
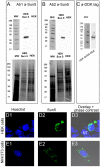
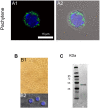
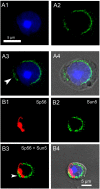
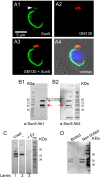
The acrosome is an organelle that is central to sperm physiology and a defective acrosome biogenesis leads to globozoospermia, a severe male infertility. The identification of the actors involved in acrosome biogenesis is therefore particularly important to decipher the molecular pathogeny of globozoospermia. We recently showed that a defect in the DPY19L2 gene is present in more than 70% of globozoospermic men and demonstrated that Dpy19l2, located in the inner nuclear membrane, is the first protein involved in the attachment of the acrosome to the nuclear envelope (NE). SUN proteins serve to link the nuclear envelope to the cytoskeleton and are therefore good candidates to participate in acrosome-nucleus attachment, potentially by interacting with DPY19L2. In order to characterize new actors of acrosomal attachment, we focused on Sun5 (also called Spag4l), which is highly expressed in male germ cells, and investigated its localization during spermatogenesis. Using immunohistochemistry and Western blot experiments in mice, we showed that Sun5 transits through different cellular compartments during meiosis. In pachytene spermatocytes, it is located in a membranous compartment different to the reticulum. In round spermatids, it progresses to the Golgi and the NE before to be located to the tail/head junction in epididymal sperm. Interestingly, we demonstrate that Sun5 is not, as initially reported, facing the acrosome but is in fact excluded from this zone. Moreover, we show that in Dpy19l2 KO spermatids, upon the detachment of the acrosome, Sun5 relocalizes to the totality of the NE suggesting that the acrosome attachment excludes Sun5 from the NE facing the acrosome. Finally, Western-blot experiments demonstrate that Sun5 is glycosylated. Overall, our work, associated with other publications, strongly suggests that the attachment of the acrosome to the nucleus does not likely depend on the formation of SUN complexes.
Conflict of interest statement
Figures

Similar articles
-
Absence of Dpy19l2, a new inner nuclear membrane protein, causes globozoospermia in mice by preventing the anchoring of the acrosome to the nucleus.Development. 2012 Aug;139(16):2955-65. doi: 10.1242/dev.077982. Epub 2012 Jul 4. Development. 2012. PMID: 22764053
-
SPAG4L/SPAG4L-2 are testis-specific SUN domain proteins restricted to the apical nuclear envelope of round spermatids facing the acrosome.Mol Hum Reprod. 2011 Apr;17(4):207-18. doi: 10.1093/molehr/gaq099. Epub 2010 Dec 15. Mol Hum Reprod. 2011. PMID: 21159740
-
Subcellular localization of phospholipase Cζ in human sperm and its absence in DPY19L2-deficient sperm are consistent with its role in oocyte activation.Mol Hum Reprod. 2015 Feb;21(2):157-68. doi: 10.1093/molehr/gau098. Epub 2014 Oct 29. Mol Hum Reprod. 2015. PMID: 25354701 Free PMC article.
-
Protein expression and cell organelle behavior in spermatogenic cells.Kaibogaku Zasshi. 2001 Jun;76(3):267-79. Kaibogaku Zasshi. 2001. PMID: 11494512 Review.
-
LINCking the Nuclear Envelope to Sperm Architecture.Genes (Basel). 2021 Apr 27;12(5):658. doi: 10.3390/genes12050658. Genes (Basel). 2021. PMID: 33925685 Free PMC article. Review.
Cited by
-
The involvement of the nuclear lamina in human and rodent spermiogenesis: a systematic review.Basic Clin Androl. 2018 Jun 20;28:7. doi: 10.1186/s12610-018-0072-4. eCollection 2018. Basic Clin Androl. 2018. PMID: 29946470 Free PMC article.
-
Sperm Morphology Assessment in the Era of Intracytoplasmic Sperm Injection: Reliable Results Require Focus on Standardization, Quality Control, and Training.World J Mens Health. 2022 Jul;40(3):347-360. doi: 10.5534/wjmh.210054. Epub 2021 Jun 17. World J Mens Health. 2022. PMID: 34169687 Free PMC article. Review.
-
Ultra-structure of the sperm head-to-tail linkage complex in the absence of the spermatid-specific LINC component SPAG4.Histochem Cell Biol. 2018 Jul;150(1):49-59. doi: 10.1007/s00418-018-1668-7. Epub 2018 Apr 16. Histochem Cell Biol. 2018. PMID: 29663073
-
SUN5 Interacting With Nesprin3 Plays an Essential Role in Sperm Head-to-Tail Linkage: Research on Sun5 Gene Knockout Mice.Front Cell Dev Biol. 2021 Jun 29;9:684826. doi: 10.3389/fcell.2021.684826. eCollection 2021. Front Cell Dev Biol. 2021. PMID: 34268309 Free PMC article.
-
[Genetic analysis of three cases of acephalic spermatozoa syndrome caused by SUN5 mutation and the outcome of assisted reproductive technology].Beijing Da Xue Xue Bao Yi Xue Ban. 2021 Aug 18;53(4):803-807. doi: 10.19723/j.issn.1671-167X.2021.04.031. Beijing Da Xue Xue Bao Yi Xue Ban. 2021. PMID: 34393249 Free PMC article. Chinese.
References
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases
Research Materials

