Structure-function analysis of peptide signaling in the Clostridium perfringens Agr-like quorum sensing system
- PMID: 25777675
- PMCID: PMC4402402
- DOI: 10.1128/JB.02614-14
Structure-function analysis of peptide signaling in the Clostridium perfringens Agr-like quorum sensing system
Abstract
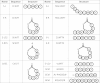
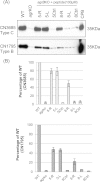
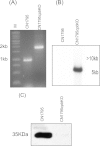
The accessory growth regulator (Agr)-like quorum sensing (QS) system of Clostridium perfringens controls the production of many toxins, including beta toxin (CPB). We previously showed (J. E. Vidal, M. Ma, J. Saputo, J. Garcia, F. A. Uzal, and B. A. McClane, Mol Microbiol 83:179-194, 2012, http://dx.doi.org/10.1111/j.1365-2958.2011.07925.x) that an 8-amino-acid, AgrD-derived peptide named 8-R upregulates CPB production by this QS system. The current study synthesized a series of small signaling peptides corresponding to sequences within the C. perfringens AgrD polypeptide to investigate the C. perfringens autoinducing peptide (AIP) structure-function relationship. When both linear and cyclic ring forms of these peptides were added to agrB null mutants of type B strain CN1795 or type C strain CN3685, the 5-amino-acid peptides, whether in a linear or ring (thiolactone or lactone) form, induced better signaling (more CPB production) than peptide 8-R for both C. perfringens strains. The 5-mer thiolactone ring peptide induced faster signaling than the 5-mer linear peptide. Strain-related variations in sensing these peptides were detected, with CN3685 sensing the synthetic peptides more strongly than CN1795. Consistent with those synthetic peptide results, Transwell coculture experiments showed that CN3685 exquisitely senses native AIP signals from other isolates (types A, B, C, and D), while CN1795 barely senses even its own AIP. Finally, a C. perfringens AgrD sequence-based peptide with a 6-amino-acid thiolactone ring interfered with CPB production by several C. perfringens strains, suggesting potential therapeutic applications. These results indicate that AIP signaling sensitivity and responsiveness vary among C. perfringens strains and suggest C. perfringens prefers a 5-mer AIP to initiate Agr signaling.
Importance: Clostridium perfringens possesses an Agr-like quorum sensing (QS) system that regulates virulence, sporulation, and toxin production. The current study used synthetic peptides to identify the structure-function relationship for the signaling peptide that activates this QS system. We found that a 5-mer peptide induces optimal signaling. Unlike other Agr systems, a linear version of this peptide (in addition to thiolactone and lactone versions) could induce signaling. Two C. perfringens strains were found to vary in sensitivity to these peptides. We also found that a 6-mer peptide can inhibit toxin production by some strains, suggesting therapeutic applications.
Copyright © 2015, American Society for Microbiology. All Rights Reserved.
Figures





Similar articles
-
The Agr-Like Quorum-Sensing System Is Important for Clostridium perfringens Type A Strain ATCC 3624 To Cause Gas Gangrene in a Mouse Model.mSphere. 2020 Jun 17;5(3):e00500-20. doi: 10.1128/mSphere.00500-20. mSphere. 2020. PMID: 32554714 Free PMC article.
-
Evidence That VirS Is a Receptor for the Signaling Peptide of the Clostridium perfringens Agr-like Quorum Sensing System.mBio. 2020 Sep 15;11(5):e02219-20. doi: 10.1128/mBio.02219-20. mBio. 2020. PMID: 32934089 Free PMC article.
-
Role of the Agr-like quorum-sensing system in regulating toxin production by Clostridium perfringens type B strains CN1793 and CN1795.Infect Immun. 2012 Sep;80(9):3008-17. doi: 10.1128/IAI.00438-12. Epub 2012 Jun 11. Infect Immun. 2012. PMID: 22689820 Free PMC article.
-
Host cell-induced signaling causes Clostridium perfringens to upregulate production of toxins important for intestinal infections.Gut Microbes. 2014 Jan-Feb;5(1):96-107. doi: 10.4161/gmic.26419. Epub 2013 Sep 10. Gut Microbes. 2014. PMID: 24061146 Free PMC article. Review.
-
Making sense of quorum sensing in lactobacilli: a special focus on Lactobacillus plantarum WCFS1.Microbiology (Reading). 2007 Dec;153(Pt 12):3939-3947. doi: 10.1099/mic.0.2007/012831-0. Microbiology (Reading). 2007. PMID: 18048908 Review.
Cited by
-
The Agr-Like Quorum-Sensing System Is Important for Clostridium perfringens Type A Strain ATCC 3624 To Cause Gas Gangrene in a Mouse Model.mSphere. 2020 Jun 17;5(3):e00500-20. doi: 10.1128/mSphere.00500-20. mSphere. 2020. PMID: 32554714 Free PMC article.
-
Autoinducing peptides regulate antibiotic production to potentially shape root microbiome.Sci Adv. 2025 Jun 20;11(25):eadw5076. doi: 10.1126/sciadv.adw5076. Epub 2025 Jun 20. Sci Adv. 2025. PMID: 40540565 Free PMC article.
-
New Weapons to Fight Old Enemies: Novel Strategies for the (Bio)control of Bacterial Biofilms in the Food Industry.Front Microbiol. 2016 Oct 18;7:1641. doi: 10.3389/fmicb.2016.01641. eCollection 2016. Front Microbiol. 2016. PMID: 27803696 Free PMC article. Review.
-
Reevaluation of whether a Functional Agr-like Quorum-Sensing System Is Necessary for Production of Wild-Type Levels of Epsilon-Toxin by Clostridium perfringens Type D Strains.mBio. 2022 Apr 26;13(2):e0049622. doi: 10.1128/mbio.00496-22. Epub 2022 Mar 23. mBio. 2022. PMID: 35319233 Free PMC article.
-
Understanding Quorum-Sensing and Biofilm Forming in Anaerobic Bacterial Communities.Int J Mol Sci. 2024 Nov 28;25(23):12808. doi: 10.3390/ijms252312808. Int J Mol Sci. 2024. PMID: 39684519 Free PMC article. Review.
References
-
- McClane BA, Uzal FA, Miyakawa MF, Lyerly D, Wilkins TD. 2006. The enterotoxic clostridia, p 688–752. In Dworkin M, Falkow S, Rosenburg E, Schleifer H, Stackebrandt E (ed), The prokaryotes, 3rd ed Springer Press, New York, NY.
-
- Rood JI. 2006. Clostridium perfringens and histotoxic disease, p 753–770. In Dworkin M, Falkow S, Rosenburg E, Schleifer H, Stackebrandt E (ed), The prokaryotes, 3rd ed Springer Press, New York, NY.
-
- McDonel JL. 1986. Toxins of Clostridium perfringens types A, B, C, D, and E, p 477–517. In Dorner F, Drews H (ed), Pharmacology of bacterial toxins. Pergamon Press, Oxford, United Kingdom.
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Miscellaneous

