A cell-penetrating ester of the neural metabolite lanthionine ketimine stimulates autophagy through the mTORC1 pathway: Evidence for a mechanism of action with pharmacological implications for neurodegenerative pathologies
- PMID: 25779968
- PMCID: PMC4569546
- DOI: 10.1016/j.nbd.2015.03.007
A cell-penetrating ester of the neural metabolite lanthionine ketimine stimulates autophagy through the mTORC1 pathway: Evidence for a mechanism of action with pharmacological implications for neurodegenerative pathologies
Abstract
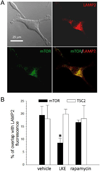
Autophagy is a fundamental cellular recycling process vulnerable to compromise in neurodegeneration. We now report that a cell-penetrating neurotrophic and neuroprotective derivative of the central nervous system (CNS) metabolite, lanthionine ketimine (LK), stimulates autophagy in RG2 glioma and SH-SY5Y neuroblastoma cells at concentrations within or below pharmacological levels reported in previous mouse studies. Autophagy stimulation was evidenced by increased lipidation of microtubule-associated protein 1 light chain 3 (LC3) both in the absence and presence of bafilomycin-A1 which discriminates between effects on autophagic flux versus blockage of autophagy clearance. LKE treatment caused changes in protein level or phosphorylation state of multiple autophagy pathway proteins including mTOR; p70S6 kinase; unc-51-like-kinase-1 (ULK1); beclin-1 and LC3 in a manner essentially identical to effects observed after rapamycin treatment. The LKE site of action was near mTOR because neither LKE nor the mTOR inhibitor rapamycin affected tuberous sclerosis complex (TSC) phosphorylation status upstream from mTOR. Confocal immunofluorescence imaging revealed that LKE specifically decreased mTOR (but not TSC2) colocalization with LAMP2(+) lysosomes in RG2 cells, a necessary event for mTORC1-mediated autophagy suppression, whereas rapamycin had no effect. Suppression of the LK-binding adaptor protein CRMP2 (collapsin response mediator protein-2) by means of shRNA resulted in diminished autophagy flux, suggesting that the LKE action on mTOR localization may occur through a novel mechanism involving CRMP2-mediated intracellular trafficking. These findings clarify the mechanism-of-action for LKE in preclinical models of CNS disease, while suggesting possible roles for natural lanthionine metabolites in regulating CNS autophagy.
Keywords: Autophagy; CRMP2; DPYSL2; Lanthionine ketimine; mTOR complex (mTORC).
Copyright © 2015 Elsevier Inc. All rights reserved.
Figures



Similar articles
-
Lanthionine ketimine ethyl ester partially rescues neurodevelopmental defects in unc-33 (DPYSL2/CRMP2) mutants.J Neurosci Res. 2013 Sep;91(9):1183-90. doi: 10.1002/jnr.23239. Epub 2013 Jul 3. J Neurosci Res. 2013. PMID: 23825043
-
Lanthionine ketimine ester provides benefit in a mouse model of multiple sclerosis.J Neurochem. 2015 Jul;134(2):302-14. doi: 10.1111/jnc.13114. Epub 2015 Apr 22. J Neurochem. 2015. PMID: 25846048
-
Proteomic identification of binding partners for the brain metabolite lanthionine ketimine (LK) and documentation of LK effects on microglia and motoneuron cell cultures.J Neurosci. 2010 Feb 24;30(8):2979-88. doi: 10.1523/JNEUROSCI.5247-09.2010. J Neurosci. 2010. PMID: 20181595 Free PMC article.
-
Mammalian target of rapamycin and tuberous sclerosis complex.J Dermatol Sci. 2015 Aug;79(2):93-100. doi: 10.1016/j.jdermsci.2015.04.005. Epub 2015 Apr 25. J Dermatol Sci. 2015. PMID: 26051878 Review.
-
Cellular and molecular effects of the mTOR inhibitor everolimus.Clin Sci (Lond). 2015 Nov;129(10):895-914. doi: 10.1042/CS20150149. Clin Sci (Lond). 2015. PMID: 26330617 Review.
Cited by
-
The Sulfur Metabolite Lanthionine: Evidence for a Role as a Novel Uremic Toxin.Toxins (Basel). 2017 Jan 10;9(1):26. doi: 10.3390/toxins9010026. Toxins (Basel). 2017. PMID: 28075397 Free PMC article. Review.
-
Impaired Autophagy in Motor Neurons: A Final Common Mechanism of Injury and Death.Physiology (Bethesda). 2018 May 1;33(3):211-224. doi: 10.1152/physiol.00008.2018. Physiology (Bethesda). 2018. PMID: 29638184 Free PMC article. Review.
-
Collapsin Response Mediator Protein-2 (CRMP2) is a Plausible Etiological Factor and Potential Therapeutic Target in Alzheimer's Disease: Comparison and Contrast with Microtubule-Associated Protein Tau.J Alzheimers Dis. 2016 Apr 15;53(1):1-14. doi: 10.3233/JAD-160076. J Alzheimers Dis. 2016. PMID: 27079722 Free PMC article. Review.
-
CTB-targeted protocells enhance ability of lanthionine ketenamine analogs to induce autophagy in motor neuron-like cells.Sci Rep. 2023 Feb 13;13(1):2581. doi: 10.1038/s41598-023-29437-8. Sci Rep. 2023. PMID: 36781993 Free PMC article.
-
An overview of sulfur-containing compounds originating from natural metabolites: Lanthionine ketimine and its analogues.Anal Biochem. 2020 Feb 15;591:113543. doi: 10.1016/j.ab.2019.113543. Epub 2019 Dec 17. Anal Biochem. 2020. PMID: 31862405 Free PMC article. Review.
References
-
- Bach M, Larance M, James DE, Ramm G. The serine/threonine kinase ULK1 is a target of multiple phosphorylation events. Biochem. J. 2011;440:283–291. - PubMed
-
- Bibee KP, Cheng YJ, Ching JK, Marsh JN, Li AJ, Keeling RM, Connolly AM, Golumbek PT, Myerson JW, Hu G, Chen J, Shannon WD, Lanza GM, Weihl CC, Wickline SA. Rapamycin nanoparticles target defective autophagy in muscular dystrophy to enhance both strength and cardiac function. FASEB J. 2014;28:2047–2061. - PMC - PubMed
-
- Cavallini D, Ricci G, Dupre S, Pecci L, Costa M, Matarese RM, Pensa B, Antonuci A, Solinas SP, Fontana M. Sulfur-containing cyclic ketimines and imino acids. A novel family of endogenous products in search for a role. Eur. J. Biochem. 1991;202:217–223. - PubMed
-
- Chiang GG, Abraham RT. Phosphorylation of mammalian target of rapamycin (mTOR) at Ser-2448 is mediated by p70S6 kinase. J. Biol. Chem. 2005;280:25485–25490. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials
Miscellaneous

