The activation of the atypical PKC zeta in light-induced retinal degeneration and its involvement in L-DNase II control
- PMID: 25781645
- PMCID: PMC4511362
- DOI: 10.1111/jcmm.12539
The activation of the atypical PKC zeta in light-induced retinal degeneration and its involvement in L-DNase II control
Abstract
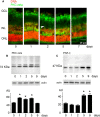
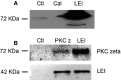
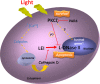
Light-induced retinal degeneration is characterized by photoreceptor cell death. Many studies showed that photoreceptor demise is caspase-independent. In our laboratory we showed that leucocyte elastase inhibitor/LEI-derived DNase II (LEI/L-DNase II), a caspase-independent apoptotic pathway, is responsible for photoreceptor death. In this work, we investigated the activation of a pro-survival kinase, the protein kinase C (PKC) zeta. We show that light exposure induced PKC zeta activation. PKC zeta interacts with LEI/L-DNase II and controls its DNase activity by impairing its nuclear translocation. These results highlight the role of PKC zeta in retinal physiology and show that this kinase can control caspase-independent pathways.
Keywords: PKC zeta; light; retinal apoptosis; retinal degeneration; serpin B1.
© 2015 The Authors. Journal of Cellular and Molecular Medicine published by John Wiley & Sons Ltd and Foundation for Cellular and Molecular Medicine.
Figures



Similar articles
-
The LEI/L-DNase II pathway is activated in light-induced retinal degeneration in rats.Neurosci Lett. 2004 Sep 2;367(2):205-9. doi: 10.1016/j.neulet.2004.06.004. Neurosci Lett. 2004. PMID: 15331154
-
Regulation of poly(ADP-ribose) polymerase-1 functions by leukocyte elastase inhibitor/LEI-derived DNase II during caspase-independent apoptosis.Int J Biochem Cell Biol. 2009 May;41(5):1046-54. doi: 10.1016/j.biocel.2008.09.026. Epub 2008 Oct 4. Int J Biochem Cell Biol. 2009. PMID: 18951996
-
Apoptosis induced by Na+/H+ antiport inhibition activates the LEI/L-DNase II pathway.Cell Death Differ. 2003 May;10(5):548-57. doi: 10.1038/sj.cdd.4401195. Cell Death Differ. 2003. PMID: 12728253
-
A caspase-independent cell clearance program. The LEI/L-DNase II pathway.Ann N Y Acad Sci. 2000;926:192-203. doi: 10.1111/j.1749-6632.2000.tb05612.x. Ann N Y Acad Sci. 2000. PMID: 11193035 Review.
-
Molecular mechanism of L-DNase II activation and function as a molecular switch in apoptosis.Biochem Pharmacol. 2008 Dec 1;76(11):1490-502. doi: 10.1016/j.bcp.2008.07.039. Epub 2008 Aug 9. Biochem Pharmacol. 2008. PMID: 18761000 Review.
Cited by
-
The hidden side of SERPINB1/Leukocyte Elastase Inhibitor.Semin Cell Dev Biol. 2017 Feb;62:178-186. doi: 10.1016/j.semcdb.2016.07.010. Epub 2016 Jul 12. Semin Cell Dev Biol. 2017. PMID: 27422329 Free PMC article. Review.
-
Animal Models of LED-Induced Phototoxicity. Short- and Long-Term In Vivo and Ex Vivo Retinal Alterations.Life (Basel). 2021 Oct 26;11(11):1137. doi: 10.3390/life11111137. Life (Basel). 2021. PMID: 34833013 Free PMC article. Review.
-
Phototoxicity of low doses of light and influence of the spectral composition on human RPE cells.Sci Rep. 2024 Mar 21;14(1):6839. doi: 10.1038/s41598-024-56980-9. Sci Rep. 2024. PMID: 38514646 Free PMC article.
-
Atypical Protein Kinase C: Breaking Down Barriers in Ocular Disease?Am J Pathol. 2018 Oct;188(10):2142-2146. doi: 10.1016/j.ajpath.2018.07.006. Epub 2018 Sep 13. Am J Pathol. 2018. PMID: 30220553 Free PMC article.
-
Effects of white light-emitting diode (LED) exposure on retinal pigment epithelium in vivo.J Cell Mol Med. 2017 Dec;21(12):3453-3466. doi: 10.1111/jcmm.13255. Epub 2017 Jun 29. J Cell Mol Med. 2017. PMID: 28661040 Free PMC article.
References
-
- Noell WK, Walker VS, Kang BS, et al. Retinal damage by light in rats. Invest Ophthalmol. 1966;5:450–73. - PubMed
-
- Organisciak DT, Darrow RA, Barsalou L, et al. Light-induced damage in the retina: differential effects of dimethylthiourea on photoreceptor survival, apoptosis and DNA oxidation. Photochem Photobiol. 1999;70:261–8. - PubMed
-
- Reme CE, Grimm C, Hafezi F, et al. Apoptotic cell death in retinal degenerations. Prog Retin Eye Res. 1998;17:443–64. - PubMed
-
- Donovan M, Carmody RJ, Cotter TG. Light-induced photoreceptor apoptosis in vivo requires neuronal nitric-oxide synthase and guanylate cyclase activity and is caspase-3-independent. J Biol Chem. 2001;276:23000–8. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases

