Thermally induced osteocyte damage initiates a remodelling signaling cascade
- PMID: 25785846
- PMCID: PMC4364670
- DOI: 10.1371/journal.pone.0119652
Thermally induced osteocyte damage initiates a remodelling signaling cascade
Abstract
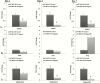
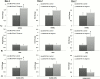
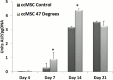
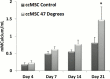
Thermal elevations experienced by bone during orthopaedic procedures, such as cutting and drilling, exothermal reactions from bone cement, and thermal therapies such as tumor ablation, can result in thermal damage leading to death of native bone cells (osteocytes, osteoblasts, osteoclasts and mesenchymal stem cells). Osteocytes are believed to be the orchestrators of bone remodeling, which recruit nearby osteoclast and osteoblasts to control resorption and bone growth in response to mechanical stimuli and physical damage. However, whether heat-induced osteocyte damage can directly elicit bone remodelling has yet to be determined. This study establishes the link between osteocyte thermal damage and the remodeling cascade. We show that osteocytes directly exposed to thermal elevations (47°C for 1 minute) become significantly apoptotic and alter the expression of osteogenic genes (Opg and Cox2). The Rankl/Opg ratio is consistently down-regulated, at days 1, 3 and 7 in MLO-Y4s heat-treated to 47°C for 1 minute. Additionally, the pro-osteoblastogenic signaling marker Cox2 is significantly up-regulated in heat-treated MLO-Y4s by day 7. Furthermore, secreted factors from heat-treated MLO-Y4s administered to MSCs using a novel co-culture system are shown to activate pre-osteoblastic MSCs to increase production of the pro-osteoblastic differentiation marker, alkaline phosphatase (day 7, 14), and calcium deposition (day 21). Most interestingly, an initial pro-osteoclastogenic signaling response (increase Rankl and Rankl/Opg ratio at day 1) followed by later stage pro-osteoblastogenic signaling (down-regulation in Rankl and the Rankl/Opg ratio and an up-regulation in Opg and Cox2 by day 7) was observed in non-heat-treated MLO-Y4s in co-culture when these were exposed to the biochemicals produced by heat-treated MLO-Y4s. Taken together, these results elucidate the vital role of osteocytes in detecting and responding to thermal damage by means of thermally induced apoptosis followed by a cascade of remodelling responses.
Conflict of interest statement
Figures



Similar articles
-
Thermally induced osteocyte damage initiates pro-osteoclastogenic gene expression in vivo.J R Soc Interface. 2016 Jun;13(119):20160337. doi: 10.1098/rsif.2016.0337. J R Soc Interface. 2016. PMID: 27335224 Free PMC article.
-
Heat-shock-induced cellular responses to temperature elevations occurring during orthopaedic cutting.J R Soc Interface. 2012 Dec 7;9(77):3503-13. doi: 10.1098/rsif.2012.0520. Epub 2012 Aug 22. J R Soc Interface. 2012. PMID: 22915633 Free PMC article.
-
Iron overload induced osteocytes apoptosis and led to bone loss in Hepcidin-/- mice through increasing sclerostin and RANKL/OPG.Bone. 2022 Nov;164:116511. doi: 10.1016/j.bone.2022.116511. Epub 2022 Aug 4. Bone. 2022. PMID: 35933095
-
Osteocyte control of bone remodeling: is sclerostin a key molecular coordinator of the balanced bone resorption-formation cycles?Osteoporos Int. 2014 Dec;25(12):2685-700. doi: 10.1007/s00198-014-2808-0. Epub 2014 Jul 17. Osteoporos Int. 2014. PMID: 25030653 Review.
-
Osteocyte-driven bone remodeling.Calcif Tissue Int. 2014 Jan;94(1):25-34. doi: 10.1007/s00223-013-9774-y. Epub 2013 Sep 4. Calcif Tissue Int. 2014. PMID: 24002178 Free PMC article. Review.
Cited by
-
Pelleted Bone Marrow Derived Mesenchymal Stem Cells Are Better Protected from the Deleterious Effects of Arthroscopic Heat Shock.Front Physiol. 2016 May 24;7:180. doi: 10.3389/fphys.2016.00180. eCollection 2016. Front Physiol. 2016. PMID: 27252654 Free PMC article.
-
Osteocyte TGFβ1‑Smad2/3 is positively associated with bone turnover parameters in subchondral bone of advanced osteoarthritis.Int J Mol Med. 2020 Jul;46(1):167-178. doi: 10.3892/ijmm.2020.4576. Epub 2020 Apr 14. Int J Mol Med. 2020. PMID: 32319543 Free PMC article.
-
How Magnetic Composites are Effective Anticancer Therapeutics? A Comprehensive Review of the Literature.Int J Nanomedicine. 2023 Jun 30;18:3535-3575. doi: 10.2147/IJN.S375964. eCollection 2023. Int J Nanomedicine. 2023. PMID: 37409027 Free PMC article. Review.
-
The Effect of TBB, as an Initiator, on the Biological Compatibility of PMMA/MMA Bone Cement.Int J Mol Sci. 2020 Jun 4;21(11):4016. doi: 10.3390/ijms21114016. Int J Mol Sci. 2020. PMID: 32512780 Free PMC article.
-
Osteocyte Apoptosis Contributes to Cold Exposure-induced Bone Loss.Front Bioeng Biotechnol. 2021 Nov 11;9:733582. doi: 10.3389/fbioe.2021.733582. eCollection 2021. Front Bioeng Biotechnol. 2021. PMID: 34858954 Free PMC article.
References
-
- Eriksson AR, Albrektsson T, Albrektsson B (1984. b) Heat caused by drilling cortical bone. Temperature measured in vivo in patients and animals. Acta Orthop Scand 55: 629–631. - PubMed
-
- Matthews LS, Hirsch C (1972) Temperatures Measured in Human Cortical Bone when Drilling. The Journal of Bone & Joint Surgery 54: 297–308. - PubMed
-
- Toksvig-Larsen S, Ryd L, Lindstrand A (1991) On the problem of heat generation in bone cutting. Studies on the effects on liquid cooling. Journal of Bone & Joint Surgery, British Volume 73-B: 13–15. - PubMed
-
- Berman AT, Reid JS, Yanicko DR, Jr., Sih GC, Zimmerman MR (1984) Thermally induced bone necrosis in rabbits. Relation to implant failure in humans. Clin Orthop Relat Res: 284–292. - PubMed
-
- Stanczyk M, van Rietbergen B (2004) Thermal analysis of bone cement polymerisation at the cement-bone interface. J Biomech 37: 1803–1810. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials

