Inflammation Combined with Ischemia Produces Myelin Injury and Plaque-Like Aggregates of Myelin, Amyloid-β and AβPP in Adult Rat Brain
- PMID: 25790832
- PMCID: PMC4878315
- DOI: 10.3233/JAD-143072
Inflammation Combined with Ischemia Produces Myelin Injury and Plaque-Like Aggregates of Myelin, Amyloid-β and AβPP in Adult Rat Brain
Abstract
Background: Ischemia, white matter injury, and Alzheimer's disease (AD) pathologies often co-exist in aging brain. How one condition predisposes to, interacts with, or perhaps causes the others remains unclear.
Objectives: To better understand the link between ischemia, white matter injury, and AD, adult rats were administered lipopolysaccharide (LPS) to serve as an inflammatory stimulus, and 24 h later subjected to 20-min focal cerebral ischemia (IS) followed by 30-min hypoxia (H).
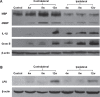
Methods: Myelin and axonal damage, as well as amyloid-β (Aβ) and amyloid-β protein precursor (AβPP) deposition were examined by Western blot and immunocytochemistry following LPS/IS/H. Findings were compared to the 5XFAD mouse AD brain.
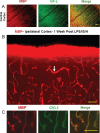
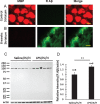
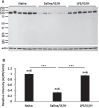
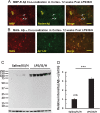
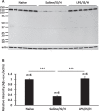
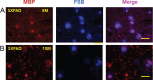
Results: Myelin/axonal injury was observed bilaterally in cortex following LPS/IS/H, along with an increase in IL-1, granzyme B, and LPS. AβPP deposition was present in ischemic striatum in regions of myelin loss. Aβ(1-42) and AβPP were deposited in small foci in ischemic cortex that co-localized with myelin aggregates. In the 5XFAD mouse AD model, cortical amyloid plaques also co-localized with myelin aggregates.
Conclusions: LPS/IS/H produce myelin injury and plaque-like aggregates of myelin. AβPP and Aβ co-localize with these myelin aggregates.
Keywords: Alzheimer’s disease; amyloid plaques; amyloid-β; amyloid-β protein precursor; hypoxia; lipopolysaccharide; myelin; myelin basic protein.
Figures




References
-
- Schneider JA, Arvanitakis Z, Bang W, Bennett DA. Mixed brain pathologies account for most dementia cases in community-dwelling older persons. Neurology. 2007;69:2197–2204. - PubMed
-
- Pohjasvaara T, Erkinjuntti T, Ylikoski R, Hietanen M, Vataja R, Kaste M. Clinical determinants of poststroke dementia. Stroke. 1998;29:75–81. - PubMed
-
- Desmond DW, Moroney JT, Paik MC, Sano M, Mohr JP, Aboumatar S, Tseng CL, Chan S, Williams JB, Remien RH, Hauser WA, Stern Y. Frequency and clinical determinants of dementia after ischemic stroke. Neurology. 2000;54:1124–1131. - PubMed
-
- Henon H, Durieu I, Guerouaou D, Lebert F, Pasquier F, Leys D. Poststroke dementia: Incidence and relationship to prestroke cognitive decline. Neurology. 2001;57:1216–1222. - PubMed

