Primary screening for cervical cancer based on high-risk human papillomavirus (HPV) detection and HPV 16 and HPV 18 genotyping, in comparison to cytology
- PMID: 25793281
- PMCID: PMC4368762
- DOI: 10.1371/journal.pone.0119755
Primary screening for cervical cancer based on high-risk human papillomavirus (HPV) detection and HPV 16 and HPV 18 genotyping, in comparison to cytology
Abstract
Objectives: The objective of the present study is to assess the performance of a high-risk human papillomavirus (HR-HPV) DNA test with individual HPV-16/HPV-18 genotyping as a method for primary cervical cancer screening compared with liquid-based cytology (LBC) in a population of Greek women taking part in routine cervical cancer screening.
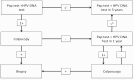
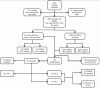
Methods: The study, conducted by the "HEllenic Real life Multicentric cErvical Screening" (HERMES) study group, involved the recruitment of 4,009 women, aged 25-55, who took part in routine cervical screening at nine Gynecology Departments in Greece. At first visit cervical specimens were collected for LBC and HPV testing using the Roche Cobas 4800 system. Women found positive for either cytology or HPV were referred for colposcopy, whereas women negative for both tests will be retested after three years. The study is ongoing and the results of the first screening round are reported herein.
Results: Valid results for cytology and HPV testing were obtained for 3,993 women. The overall prevalence of HR-HPV was 12.7%, of HPV-16 2.7% and of HPV-18 1.4%. Of those referred for colposcopy, cervical intraepithelial neoplasia grade 2 or worse (CIN2+) was detected in 41 women (1.07%). At the threshold of CIN2+, cytology [atypical squamous cells of undetermined significance (ASC-US) or worse] and HPV testing showed a sensitivity of 53.7% and 100% respectively, without change between age groups. Cytology and HPV testing showed specificity of 96.8% and 90.3% respectively, which was increased in older women (≥30) in comparison to younger ones (25-29). Genotyping for HPV16/18 had similar accuracy to cytology for the detection of CIN2+ (sensitivity: 58.5%; specificity 97.5%) as well as for triage to colposcopy (sensitivity: 58.5% vs 53.7% for cytology).
Conclusion: HPV testing has much better sensitivity than cytology to identify high-grade cervical lesions with slightly lower specificity. HPV testing with individual HPV-16/HPV-18 genotyping could represent a more accurate methodology for primary cervical cancer screening in comparison to liquid-based cytology, especially in older women.
Conflict of interest statement
Figures
Similar articles
-
Performance of carcinogenic human papillomavirus (HPV) testing and HPV16 or HPV18 genotyping for cervical cancer screening of women aged 25 years and older: a subanalysis of the ATHENA study.Lancet Oncol. 2011 Sep;12(9):880-90. doi: 10.1016/S1470-2045(11)70188-7. Epub 2011 Aug 22. Lancet Oncol. 2011. PMID: 21865084 Clinical Trial.
-
Comparison of HPV-16 and HPV-18 Genotyping and Cytological Testing as Triage Testing Within Human Papillomavirus-Based Screening in Mexico.JAMA Netw Open. 2019 Nov 1;2(11):e1915781. doi: 10.1001/jamanetworkopen.2019.15781. JAMA Netw Open. 2019. PMID: 31747033 Free PMC article.
-
Cervical screening with primary HPV testing or cytology in a population of women in which those aged 33 years or younger had previously been offered HPV vaccination: Results of the Compass pilot randomised trial.PLoS Med. 2017 Sep 19;14(9):e1002388. doi: 10.1371/journal.pmed.1002388. eCollection 2017 Sep. PLoS Med. 2017. PMID: 28926579 Free PMC article. Clinical Trial.
-
Genotyping for Human Papillomavirus Types 16 and 18 in Women With Minor Cervical Lesions: A Systematic Review and Meta-analysis.Ann Intern Med. 2017 Jan 17;166(2):118-127. doi: 10.7326/M15-2735. Epub 2016 Nov 15. Ann Intern Med. 2017. PMID: 27842420
-
Evidence regarding human papillomavirus testing in secondary prevention of cervical cancer.Vaccine. 2012 Nov 20;30 Suppl 5:F88-99. doi: 10.1016/j.vaccine.2012.06.095. Vaccine. 2012. PMID: 23199969 Review.
Cited by
-
Efficacy of careHPV™ human papillomavirus screening versus conventional cytology tests for the detection of precancerous and cancerous cervical lesions among women living with HIV-1 in Lao People's Democratic Republic.Cancer Med. 2022 May;11(9):1984-1994. doi: 10.1002/cam4.4502. Epub 2022 Mar 8. Cancer Med. 2022. PMID: 35257506 Free PMC article.
-
Reporting of test comparisons in diagnostic accuracy studies: A literature review.Res Synth Methods. 2021 May;12(3):357-367. doi: 10.1002/jrsm.1469. Epub 2020 Dec 10. Res Synth Methods. 2021. PMID: 33217225 Free PMC article. Review.
-
HPV Testing for Cervical Cancer in Romania: High-Risk HPV Prevalence among Ethnic Subpopulations and Regions.Ann Glob Health. 2019 Jun 20;85(1):89. doi: 10.5334/aogh.2502. Ann Glob Health. 2019. PMID: 31225959 Free PMC article.
-
Evaluation of silica spin‑column and magnetic bead formats for rapid DNA methylation analysis in clinical and point‑of‑care settings.Biomed Rep. 2024 Jun 10;21(2):112. doi: 10.3892/br.2024.1800. eCollection 2024 Aug. Biomed Rep. 2024. PMID: 38912171 Free PMC article.
-
The p16/ki-67 assay is a safe, effective and rapid approach to triage women with mild cervical lesions.PLoS One. 2021 Jun 11;16(6):e0253045. doi: 10.1371/journal.pone.0253045. eCollection 2021. PLoS One. 2021. PMID: 34115809 Free PMC article.
References
-
- Koss LG The Papanicolaou test for cervical cancer detection. A triumph and a tragedy. JAMA. 1989;261: 737–743. - PubMed
-
- Bosch FX, Munoz N, Shah KV, Meheus A. Second International Workshop on the Epidemiology of Cervical Cancer and Human Papillomaviruses. Int J Cancer. 1992;52: 171–173. - PubMed
-
- zur Hausen H. Human papillomaviruses and their possible role in squamous cell carcinomas. Curr Top Microbiol Immunol. 1977;78: 1–30. - PubMed
MeSH terms
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Miscellaneous