The PKD inhibitor CID755673 enhances cardiac function in diabetic db/db mice
- PMID: 25798941
- PMCID: PMC4370864
- DOI: 10.1371/journal.pone.0120934
The PKD inhibitor CID755673 enhances cardiac function in diabetic db/db mice
Abstract
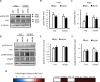
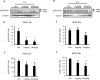
The development of diabetic cardiomyopathy is a key contributor to heart failure and mortality in obesity and type 2 diabetes (T2D). Current therapeutic interventions for T2D have limited impact on the development of diabetic cardiomyopathy. Clearly, new therapies are urgently needed. A potential therapeutic target is protein kinase D (PKD), which is activated by metabolic insults and implicated in the regulation of cardiac metabolism, contractility and hypertrophy. We therefore hypothesised that PKD inhibition would enhance cardiac function in T2D mice. We first validated the obese and T2D db/db mouse as a model of early stage diabetic cardiomyopathy, which was characterised by both diastolic and systolic dysfunction, without overt alterations in left ventricular morphology. These functional characteristics were also associated with increased PKD2 phosphorylation in the fed state and a gene expression signature characteristic of PKD activation. Acute administration of the PKD inhibitor CID755673 to normal mice reduced both PKD1 and 2 phosphorylation in a time and dose-dependent manner. Chronic CID755673 administration to T2D db/db mice for two weeks reduced expression of the gene expression signature of PKD activation, enhanced indices of both diastolic and systolic left ventricular function and was associated with reduced heart weight. These alterations in cardiac function were independent of changes in glucose homeostasis, insulin action and body composition. These findings suggest that PKD inhibition could be an effective strategy to enhance heart function in obese and diabetic patients and provide an impetus for further mechanistic investigations into the role of PKD in diabetic cardiomyopathy.
Conflict of interest statement
Figures




References
-
- Geiss LS, Herman WH, Smith PJ, Group NDD (1995) Diabetes in America. Bethesda, MD: National Institutes of Health, National Institute of Diabetes and Digestive and Kidney Diseases. 233–257 p.
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Molecular Biology Databases
Miscellaneous

