Validation of LRG1 as a potential biomarker for detection of epithelial ovarian cancer by a blinded study
- PMID: 25799488
- PMCID: PMC4370724
- DOI: 10.1371/journal.pone.0121112
Validation of LRG1 as a potential biomarker for detection of epithelial ovarian cancer by a blinded study
Abstract
Background: Leucine-rich alpha-2-glycoprotein (LRG1) was found to be differentially expressed in sera from patients with Epithelial Ovarian Cancer (EOC). The aim of this study is to investigate the performance of LRG1 for detection of EOC, including early stage EOC, and to evaluate if LRG1 can complement CA125 in order to improve EOC detection using two independent blinded sample sets.
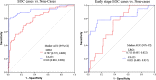
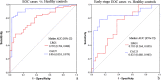
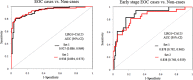
Methods and results: Serum LRG1 and CA125 were measured by immunoassays. All assays were performed blinded to clinical data. Using the two independent sample sets (156 participants for sample set 1, and 233 for sample set 2), LRG1 was differentially expressed in EOC cases as compared to healthy, surgical, and benign controls, and its performance was not affected by the conditions of blood collection. The areas under the ROC curve (AUC) for LRG1 in differentiating EOC cases from non-cases were 0.797 and 0.786 for sample set 1 and 2. For differentiating EOC cases from healthy controls, the AUC values for LRG1 were 0.792 and 0.794. At a fixed specificity of 95%, LRG1 detects 52%, and 53.5% of EOC cases from healthy controls for sample set 1 and 2. When combining LRG1 and CA125, the AUC value increased to 0.927, which was improved compared to CA125 (AUC=0.916) (p=0.008) alone in distinguishing EOC cases from non-cases. More importantly, LRG1 also showed potential performance in differentiating early stage EOC from non-cases with an AUC of 0.715 for sample set 1, and 0.690 for sample set 2. The combination of LRG1 and CA125 resulted in an AUC of 0.838, which outperforms CA125 (AUC=0.785) (p=0.018) in detecting early stage EOC cases from non-cases using the larger sample set.
Conclusions: LRG1 could be a useful biomarker alone or in combination with CA125 for the diagnosis of ovarian cancer.
Conflict of interest statement
Figures


Similar articles
-
Diagnostic Model of Serum miR-193a-5p, HE4 and CA125 Improves the Diagnostic Efficacy of Epithelium Ovarian Cancer.Pathol Oncol Res. 2018 Oct;24(4):739-744. doi: 10.1007/s12253-018-0392-x. Epub 2018 Mar 8. Pathol Oncol Res. 2018. PMID: 29520570
-
Diagnostic performance of the biomarkers HE4 and CA125 in type I and type II epithelial ovarian cancer.Gynecol Oncol. 2013 Oct;131(1):52-8. doi: 10.1016/j.ygyno.2013.07.094. Epub 2013 Jul 25. Gynecol Oncol. 2013. PMID: 23891789 Clinical Trial.
-
A multiplex biomarker assay improves the diagnostic performance of HE4 and CA125 in ovarian tumor patients.PLoS One. 2020 Oct 19;15(10):e0240418. doi: 10.1371/journal.pone.0240418. eCollection 2020. PLoS One. 2020. PMID: 33075095 Free PMC article.
-
Prognostic and predictive value of CA-125 in the primary treatment of epithelial ovarian cancer: potentials and pitfalls.Clin Transl Oncol. 2012 Jan;14(1):15-20. doi: 10.1007/s12094-012-0756-8. Clin Transl Oncol. 2012. PMID: 22262714 Review.
-
Microvesicles as potential ovarian cancer biomarkers.Biomed Res Int. 2013;2013:703048. doi: 10.1155/2013/703048. Epub 2013 Jan 8. Biomed Res Int. 2013. PMID: 23484144 Free PMC article. Review.
Cited by
-
Evaluation of a biomarker for the diagnosis of pancreas cancer using an animal model.J Toxicol Pathol. 2019 Jul;32(3):135-141. doi: 10.1293/tox.2018-0062. Epub 2019 Apr 4. J Toxicol Pathol. 2019. PMID: 31404387 Free PMC article.
-
[LRG1 inhibits hepatic macrophage activation by enhancing TGF-β1 signaling to alleviate MAFLD in mice].Nan Fang Yi Ke Da Xue Xue Bao. 2023 Jul 20;43(7):1164-1171. doi: 10.12122/j.issn.1673-4254.2023.07.13. Nan Fang Yi Ke Da Xue Xue Bao. 2023. PMID: 37488799 Free PMC article. Chinese.
-
LRG1 Suppresses Migration and Invasion of Esophageal Squamous Cell Carcinoma by Modulating Epithelial to Mesenchymal Transition.J Cancer. 2020 Jan 14;11(6):1486-1494. doi: 10.7150/jca.36189. eCollection 2020. J Cancer. 2020. PMID: 32047555 Free PMC article.
-
Circulating microvesicles correlate with radiation proctitis complication after radiotherapy.Sci Rep. 2023 Feb 4;13(1):2033. doi: 10.1038/s41598-022-21726-y. Sci Rep. 2023. PMID: 36739457 Free PMC article.
-
Research Progress on Leucine-Rich Alpha-2 Glycoprotein 1: A Review.Front Pharmacol. 2022 Jan 5;12:809225. doi: 10.3389/fphar.2021.809225. eCollection 2021. Front Pharmacol. 2022. PMID: 35095520 Free PMC article. Review.
References
-
- Holschneider CH, Berek JS. Ovarian cancer: epidemiology, biology, and prognostic factors. Semin Surg Oncol. 2000; 19: 3–10. - PubMed
-
- Woolas RP, Xu FJ, Jacobs IJ, Yu YH, Daly L, Berchuck A, et al. Elevation of multiple serum markers in patients with stage I ovarian cancer. J Natl Cancer Inst. 1993; 85: 1748–51. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Research Materials
Miscellaneous

