Circulating tumor cell biomarker panel as an individual-level surrogate for survival in metastatic castration-resistant prostate cancer
- PMID: 25800753
- PMCID: PMC4397279
- DOI: 10.1200/JCO.2014.55.3487
Circulating tumor cell biomarker panel as an individual-level surrogate for survival in metastatic castration-resistant prostate cancer
Abstract
Purpose: Trials in castration-resistant prostate cancer (CRPC) need new clinical end points that are valid surrogates for survival. We evaluated circulating tumor cell (CTC) enumeration as a surrogate outcome measure.
Patients and methods: Examining CTCs alone and in combination with other biomarkers as a surrogate for overall survival was a secondary objective of COU-AA-301, a multinational, randomized, double-blind phase III trial of abiraterone acetate plus prednisone versus prednisone alone in patients with metastatic CRPC previously treated with docetaxel. The biomarkers were measured at baseline and 4, 8, and 12 weeks, with 12 weeks being the primary measure of interest. The Prentice criteria were applied to test candidate biomarkers as surrogates for overall survival at the individual-patient level.
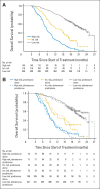
Results: A biomarker panel using CTC count and lactate dehydrogenase (LDH) level was shown to satisfy the four Prentice criteria for individual-level surrogacy. Twelve-week surrogate biomarker data were available for 711 patients. The abiraterone acetate plus prednisone and prednisone-alone groups demonstrated a significant survival difference (P = .034); surrogate distribution at 12 weeks differed by treatment (P < .001); the discriminatory power of the surrogate to predict mortality was high (weighted c-index, 0.81); and adding the surrogate to the model eliminated the treatment effect on survival. Overall, 2-year survival of patients with CTCs < 5 (low risk) versus patients with CTCs ≥ 5 cells/7.5 mL of blood and LDH > 250 U/L (high risk) at 12 weeks was 46% and 2%, respectively.
Conclusion: A biomarker panel containing CTC number and LDH level was shown to be a surrogate for survival at the individual-patient level in this trial of abiraterone acetate plus prednisone versus prednisone alone for patients with metastatic CRPC. Additional trials are ongoing to validate the findings.
Trial registration: ClinicalTrials.gov NCT00638690.
© 2015 by American Society of Clinical Oncology.
Conflict of interest statement
Authors' disclosures of potential conflicts of interest and author contributions are found at the end of this article.
Figures
Comment in
-
Making progress on progression in metastatic prostate cancer.J Clin Oncol. 2015 Apr 20;33(12):1322-4. doi: 10.1200/JCO.2014.59.4283. Epub 2015 Feb 9. J Clin Oncol. 2015. PMID: 25667271 No abstract available.
-
Prostate cancer: New biomarker panel prognosticates patient survival.Nat Rev Clin Oncol. 2015 Jun;12(6):310. doi: 10.1038/nrclinonc.2015.68. Epub 2015 Apr 7. Nat Rev Clin Oncol. 2015. PMID: 25850548 No abstract available.
-
Prostate cancer: New biomarker panel prognosticates patient survival.Nat Rev Urol. 2015 May;12(5):241. doi: 10.1038/nrurol.2015.154. Epub 2015 Apr 7. Nat Rev Urol. 2015. PMID: 25850797 No abstract available.
-
CTCs Could Shorten Drug Trials.Cancer Discov. 2015 Jun;5(6):570-1. doi: 10.1158/2159-8290.CD-NB2015-050. Epub 2015 Apr 16. Cancer Discov. 2015. PMID: 25883015
-
Reply to A. Addeo and A. Bahl.J Clin Oncol. 2016 Feb 1;34(4):387-8. doi: 10.1200/JCO.2015.64.6562. Epub 2015 Nov 23. J Clin Oncol. 2016. PMID: 26598756 No abstract available.
-
Do All High- and Intermediate-Risk Patients With Metastatic Castration-Resistant Prostate Cancer Really Benefit From Abiraterone?J Clin Oncol. 2016 Feb 1;34(4):387. doi: 10.1200/JCO.2015.62.8537. Epub 2015 Nov 23. J Clin Oncol. 2016. PMID: 26598758 No abstract available.
References
-
- de Bono JS, Oudard S, Ozguroglu M, et al. Prednisone plus cabazitaxel or mitoxantrone for metastatic castration-resistant prostate cancer progressing after docetaxel treatment: A randomised open-label trial. Lancet. 2010;376:1147–1154. - PubMed
-
- Fizazi K, Scher HI, Molina A, et al. Abiraterone acetate for treatment of metastatic castration-resistant prostate cancer: Final overall survival analysis of the COU-AA-301 randomised, double-blind, placebo-controlled phase 3 study. Lancet Oncol. 2012;13:983–992. - PubMed
-
- Kantoff PW, Higano CS, Shore ND, et al. Sipuleucel-T immunotherapy for castration-resistant prostate cancer. N Engl J Med. 2010;363:411–422. - PubMed
-
- Parker C, Nilsson S, Heinrich D, et al. Alpha emitter radium-223 and survival in metastatic prostate cancer. N Engl J Med. 2013;369:213–223. - PubMed
Publication types
MeSH terms
Substances
Associated data
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical

