Brincidofovir (CMX001) inhibits BK polyomavirus replication in primary human urothelial cells
- PMID: 25801568
- PMCID: PMC4432119
- DOI: 10.1128/AAC.00238-15
Brincidofovir (CMX001) inhibits BK polyomavirus replication in primary human urothelial cells
Abstract
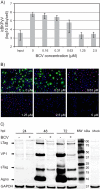
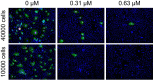
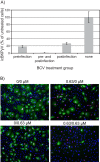
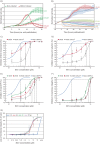
BK polyomavirus (BKPyV)-associated hemorrhagic cystitis (PyVHC) complicates 5 to 15% of allogeneic hematopoietic stem cell transplantations. Targeted antivirals are still unavailable. Brincidofovir (BCV; previously CMX001) has shown inhibitory activity against diverse viruses, including BKPyV in a primary human renal tubule cell culture model of polyomavirus-associated nephropathy. We investigated the effects of BCV in BKPyV-infected and uninfected primary human urothelial cells (HUCs), the target cells of BKPyV in PyVHC. The BCV concentrations causing 50 and 90% reductions (EC50 and EC90) in the number of intracellular BKPyV genome equivalents per cell (icBKPyV) were 0.27 μM and 0.59 μM, respectively. At 0.63 μM, BCV reduced viral late gene expression by 90% and halted progeny release. Preinfection treatment for only 24 h reduced icBKPyV similarly to treatment from 2 to 72 h postinfection, while combined pre- and postinfection treatment suppressed icBKPyV completely. After investigating BCV's effects on HUC viability, mean selectivity indices at 50 and 90% inhibition (SI50 and SI90) calculated for cellular DNA replication were 2.7 and 2.9, respectively, those for mitochondrial activity were 8.9 and 10.4, those for total ATP were 8.6 and 8.2, and those for membrane integrity were 25.9 and 16.7. The antiviral and cytostatic effects, but less so the cytotoxic effects, were inversely related to cell density. The cytotoxic effects at concentrations of ≥10 μM were rapid and likely related to BCV's lipid moiety. After carefully defining the antiviral, cytostatic, and cytotoxic properties of BCV in HUCs, we conclude that a preemptive or prophylactic approach in PyVHC is likely to give the best results.
Copyright © 2015, American Society for Microbiology. All Rights Reserved.
Figures

Similar articles
-
1-O-hexadecyloxypropyl cidofovir (CMX001) effectively inhibits polyomavirus BK replication in primary human renal tubular epithelial cells.Antimicrob Agents Chemother. 2010 Nov;54(11):4714-22. doi: 10.1128/AAC.00974-10. Epub 2010 Aug 16. Antimicrob Agents Chemother. 2010. PMID: 20713664 Free PMC article.
-
Acitretin and Retinoic Acid Derivatives Inhibit BK Polyomavirus Replication in Primary Human Proximal Renal Tubular Epithelial and Urothelial Cells.J Virol. 2021 Jul 12;95(15):e0012721. doi: 10.1128/JVI.00127-21. Epub 2021 Jul 12. J Virol. 2021. PMID: 34011542 Free PMC article.
-
Effect of Leflunomide, Cidofovir and Ciprofloxacin on replication of BKPyV in a salivary gland in vitro culture system.Antiviral Res. 2015 Jun;118:46-55. doi: 10.1016/j.antiviral.2015.02.002. Epub 2015 Mar 16. Antiviral Res. 2015. PMID: 25790744 Free PMC article.
-
Brincidofovir: understanding its unique profile and potential role against adenovirus and other viral infections.Future Microbiol. 2020 Apr;15:389-400. doi: 10.2217/fmb-2019-0288. Epub 2020 Mar 13. Future Microbiol. 2020. PMID: 32166967 Review.
-
The human polyomavirus BK (BKPyV): virological background and clinical implications.APMIS. 2013 Aug;121(8):728-45. doi: 10.1111/apm.12134. Epub 2013 Jun 19. APMIS. 2013. PMID: 23782063 Review.
Cited by
-
Presentation of BK polyomavirus-associated hemorrhagic cystitis after allogeneic hematopoietic cell transplantation.Blood Adv. 2020 Feb 25;4(4):617-628. doi: 10.1182/bloodadvances.2019000802. Blood Adv. 2020. PMID: 32074279 Free PMC article.
-
Treatment protocols for BK virus associated hemorrhagic cystitis after hematopoietic stem cell transplantation.Am J Blood Res. 2020 Oct 15;10(5):217-230. eCollection 2020. Am J Blood Res. 2020. PMID: 33224566 Free PMC article. Review.
-
Brincidofovir (CMX001) Toxicity Associated With Epithelial Apoptosis and Crypt Drop Out in a Hematopoietic Cell Transplant Patient: Challenges in Distinguishing Drug Toxicity From GVHD.J Pediatr Hematol Oncol. 2018 Aug;40(6):e364-e368. doi: 10.1097/MPH.0000000000001227. J Pediatr Hematol Oncol. 2018. PMID: 29846280 Free PMC article.
-
Antiviral Treatments.Clin Chest Med. 2017 Mar;38(1):139-153. doi: 10.1016/j.ccm.2016.11.008. Epub 2016 Dec 13. Clin Chest Med. 2017. PMID: 28159156 Free PMC article. Review.
-
Brincidofovir inhibits polyomavirus infection in vivo.mBio. 2024 Aug 14;15(8):e0104924. doi: 10.1128/mbio.01049-24. Epub 2024 Jul 2. mBio. 2024. PMID: 38953354 Free PMC article.
References
-
- Hirsch H. 2010. Polyoma and papilloma virus infections after hematopoietic stem cell or solid organ transplantation, p 465–482. In Bowden R, Ljungman P, Snydman DR (ed), Transplant infections, 3rd ed Lippincott Williams & Wilkins, Philadelphia, PA.
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources

