Multi-centre reproducibility of diffusion MRI parameters for clinical sequences in the brain
- PMID: 25802212
- PMCID: PMC4403968
- DOI: 10.1002/nbm.3269
Multi-centre reproducibility of diffusion MRI parameters for clinical sequences in the brain
Abstract
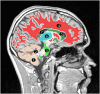
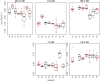
The purpose of this work was to assess the reproducibility of diffusion imaging, and in particular the apparent diffusion coefficient (ADC), intra-voxel incoherent motion (IVIM) parameters and diffusion tensor imaging (DTI) parameters, across multiple centres using clinically available protocols with limited harmonization between sequences. An ice-water phantom and nine healthy volunteers were scanned across fives centres on eight scanners (four Siemens 1.5T, four Philips 3T). The mean ADC, IVIM parameters (diffusion coefficient D and perfusion fraction f) and DTI parameters (mean diffusivity MD and fractional anisotropy FA), were measured in grey matter, white matter and specific brain sub-regions. A mixed effect model was used to measure the intra- and inter-scanner coefficient of variation (CV) for each of the five parameters. ADC, D, MD and FA had a good intra- and inter-scanner reproducibility in both grey and white matter, with a CV ranging between 1% and 7.4%; mean 2.6%. Other brain regions also showed high levels of reproducibility except for small structures such as the choroid plexus. The IVIM parameter f had a higher intra-scanner CV of 8.4% and inter-scanner CV of 24.8%. No major difference in the inter-scanner CV for ADC, D, MD and FA was observed when analysing the 1.5T and 3T scanners separately. ADC, D, MD and FA all showed good intra-scanner reproducibility, with the inter-scanner reproducibility being comparable or faring slightly worse, suggesting that using data from multiple scanners does not have an adverse effect compared with using data from the same scanner. The IVIM parameter f had a poorer inter-scanner CV when scanners of different field strengths were combined, and the parameter was also affected by the scan acquisition resolution. This study shows that the majority of diffusion MRI derived parameters are robust across 1.5T and 3T scanners and suitable for use in multi-centre clinical studies and trials.
Keywords: MRI; brain; diffusion; multi-centre; reproducibility.
© 2015 The Authors NMR in Biomedicine Published by John Wiley & Sons Ltd.
Figures





References
-
- Stejskal EO, Tanner JE. Spin diffusion measurements: spin echoes in the presence of a time-dependent field gradient. J. Chem. Phys. 1965;42:288–292. DOI: 10.1063/1.1695690. - DOI
-
- Basser PJ, Mattiello J, LeBihan D. MR diffusion tensor spectroscopy and imaging. Biophys. J. 1994;66:259–267. DOI: 10.1016/S0006-3495(94)80775-1. - DOI - PMC - PubMed
-
- Le Bihan D, Breton E, Lallemand D, Grenier P, Cabanis E, Laval-Jeantet M. MR imaging of intravoxel incoherent motions: application to diffusion and perfusion in neurologic disorders. Radiology. 1986;161:401–407. - PubMed
-
- Hales PW, Clark CA. Combined arterial spin labeling and diffusion-weighted imaging for noninvasive estimation of capillary volume fraction and permeability–surface product in the human brain. J. Cereb. Blood Flow Metab. 2013;33:67–75. DOI: 10.1038/jcbfm.2012.125. - DOI - PMC - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources

