Planteose as a storage carbohydrate required for early stage of germination of Orobanche minor and its metabolism as a possible target for selective control
- PMID: 25821071
- PMCID: PMC4449533
- DOI: 10.1093/jxb/erv116
Planteose as a storage carbohydrate required for early stage of germination of Orobanche minor and its metabolism as a possible target for selective control
Abstract
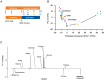
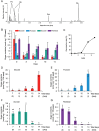
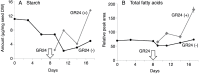
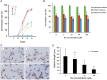
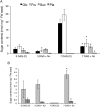
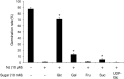
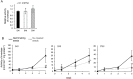
Root parasitic weeds in Orobanchaceae cause serious damage to worldwide agriculture. Germination of the parasites requires host-derived germination stimulants, such as strigolactones, as indicators of host roots within reach of the parasite's radicles. This unique germination process was focused on to identify metabolic pathways required for germination, and to design a selective control strategy. A metabolomic analysis of germinating seeds of clover broomrape, Orobanche minor, was conducted to identify its distinctive metabolites. Consequently, a galactosyl-sucrose trisaccharide, planteose (α-d-galactopyranosyl-(1→6)-β-d-fructofuranosyl-(2→1)-α-d-glucopyranoside), was identified as a metabolite that decreased promptly after reception of the germination stimulant. To investigate the importance of planteose metabolism, the effects of several glycosidase inhibitors were examined, and nojirimycin bisulfite (NJ) was found to alter the sugar metabolism and to selectively inhibit the germination of O. minor. Planteose consumption was similar in NJ-treated seeds and non-treated germinating seeds; however, NJ-treated seeds showed lower consumption of sucrose, a possible intermediate of planteose metabolism, resulting in significantly less glucose and fructose. This inhibitory effect was recovered by adding glucose. These results suggest that planteose is a storage carbohydrate required for early stage of germination of O. minor, and NJ inhibits germination by blocking the supply of essential glucose from planteose and sucrose. Additionally, NJ selectively inhibited radicle elongation of germinated seeds of Orobanchaceae plants (Striga hermonthica and Phtheirospermum japonicum). Thus, NJ will be a promising tool to develop specific herbicides to the parasites, especially broomrapes, and to improve our understanding of the molecular mechanisms of this unique germination.
Keywords: Broomrapes; metabolomics; nojirimycin; planteose; root parasitic weeds; seed germination; selective control; sugar metabolism..
© The Author 2015. Published by Oxford University Press on behalf of the Society for Experimental Biology.
Figures

References
-
- Akiyama K, Matsuzaki K, Hayashi H. 2005. Plant sesquiterpenes induce hyphal branching in arbuscular mycorrhizal fungi. Nature 435, 824–827. - PubMed
-
- Aoki T, Hatanaka S. 1991. Effect of plant growth of a sugar analogue, 1,4-dideoxy-1,4-iminoarabinitol: inhibition of invertase activity and sucrose transport in lucerne seedlings. Phytochemistry 30, 3197–3201.
-
- Aliferis KA, Chrysayi-Tokousbalides M. 2010. Metabolomics in pesticide research and development: review and future perspectives. Metabolomics 7, 35–53.
-
- Aliferis KA, Jabaji S. 2011. Metabolomics – a robust bioanalytical approach for the discovery of the modes-of-action of pesticides: a review. Pesticide Biochemistry and Physiology 100, 105–117.
-
- Aly R. 2007. Conventional and biotechnological approaches for control of parasitic weeds. In Vitro Cellular & Developmental Biology - Plant 43, 304–317.
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources

