Hydrophilic bile acids protect human blood-brain barrier endothelial cells from disruption by unconjugated bilirubin: an in vitro study
- PMID: 25821432
- PMCID: PMC4358072
- DOI: 10.3389/fnins.2015.00080
Hydrophilic bile acids protect human blood-brain barrier endothelial cells from disruption by unconjugated bilirubin: an in vitro study
Abstract
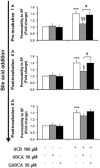
Ursodeoxycholic acid and its main conjugate glycoursodeoxycholic acid are bile acids with neuroprotective properties. Our previous studies demonstrated their anti-apoptotic, anti-inflammatory, and antioxidant properties in neural cells exposed to elevated levels of unconjugated bilirubin (UCB) as in severe jaundice. In a simplified model of the blood-brain barrier, formed by confluent monolayers of a cell line of human brain microvascular endothelial cells, UCB has shown to induce caspase-3 activation and cell death, as well as interleukin-6 release and a loss of blood-brain barrier integrity. Here, we tested the preventive and restorative effects of these bile acids regarding the disruption of blood-brain barrier properties by UCB in in vitro conditions mimicking severe neonatal hyperbilirubinemia and using the same experimental blood-brain barrier model. Both bile acids reduced the apoptotic cell death induced by UCB, but only glycoursodeoxycholic acid significantly counteracted caspase-3 activation. Bile acids also prevented the upregulation of interleukin-6 mRNA, whereas only ursodeoxycholic acid abrogated cytokine release. Regarding barrier integrity, only ursodeoxycholic acid abrogated UCB-induced barrier permeability. Better protective effects were obtained by bile acid pre-treatment, but a strong efficacy was still observed by their addition after UCB treatment. Finally, both bile acids showed ability to cross confluent monolayers of human brain microvascular endothelial cells in a time-dependent manner. Collectively, data disclose a therapeutic time-window for preventive and restorative effects of ursodeoxycholic acid and glycoursodeoxycholic acid against UCB-induced blood-brain barrier disruption and damage to human brain microvascular endothelial cells.
Keywords: blood-brain barrier; glycoursodeoxycholic acid; human brain microvascular endothelial cells; interleukin-6; unconjugated bilirubin; ursodeoxycholic acid.
Figures




Similar articles
-
Elevated levels of bilirubin and long-term exposure impair human brain microvascular endothelial cell integrity.Curr Neurovasc Res. 2011 May;8(2):153-69. doi: 10.2174/156720211795495358. Curr Neurovasc Res. 2011. PMID: 21463246
-
Glycoursodeoxycholic acid and interleukin-10 modulate the reactivity of rat cortical astrocytes to unconjugated bilirubin.J Neuropathol Exp Neurol. 2007 Sep;66(9):789-98. doi: 10.1097/nen.0b013e3181461c74. J Neuropathol Exp Neurol. 2007. PMID: 17805009
-
Neuritic growth impairment and cell death by unconjugated bilirubin is mediated by NO and glutamate, modulated by microglia, and prevented by glycoursodeoxycholic acid and interleukin-10.Neuropharmacology. 2012 Jun;62(7):2398-408. doi: 10.1016/j.neuropharm.2012.02.002. Epub 2012 Feb 13. Neuropharmacology. 2012. PMID: 22361233
-
Contribution of inflammatory processes to nerve cell toxicity by bilirubin and efficacy of potential therapeutic agents.Curr Pharm Des. 2009;15(25):2915-26. doi: 10.2174/138161209789058165. Curr Pharm Des. 2009. PMID: 19754368 Review.
-
Bilirubin chemistry and metabolism; harmful and protective aspects.Curr Pharm Des. 2009;15(25):2869-83. doi: 10.2174/138161209789058237. Curr Pharm Des. 2009. PMID: 19754364 Review.
Cited by
-
Role of ursodeoxycholic acid in neonatal indirect hyperbilirubinemia: a systematic review and meta-analysis of randomized controlled trials.Ital J Pediatr. 2022 Oct 17;48(1):179. doi: 10.1186/s13052-022-01372-w. Ital J Pediatr. 2022. PMID: 36253867 Free PMC article.
-
Reductive effect of ursodeoxycholic acid on bilirubin levels in neonates on phototherapy.Clin Exp Gastroenterol. 2019 Jul 29;12:349-354. doi: 10.2147/CEG.S207523. eCollection 2019. Clin Exp Gastroenterol. 2019. PMID: 31534356 Free PMC article.
-
The Role of Bile Acids in the Human Body and in the Development of Diseases.Molecules. 2022 May 25;27(11):3401. doi: 10.3390/molecules27113401. Molecules. 2022. PMID: 35684337 Free PMC article. Review.
-
Extensive alterations of blood metabolites in pediatric cerebral malaria.PLoS One. 2017 Apr 20;12(4):e0175686. doi: 10.1371/journal.pone.0175686. eCollection 2017. PLoS One. 2017. PMID: 28426698 Free PMC article.
-
The role of the gut microbiome and its metabolites in cerebrovascular diseases.Front Microbiol. 2023 Apr 14;14:1097148. doi: 10.3389/fmicb.2023.1097148. eCollection 2023. Front Microbiol. 2023. PMID: 37125201 Free PMC article. Review.
References
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials

