Quantitative determination of lercanidipine enantiomers in commercial formulations by capillary electrophoresis
- PMID: 25821632
- PMCID: PMC4363879
- DOI: 10.1155/2015/294270
Quantitative determination of lercanidipine enantiomers in commercial formulations by capillary electrophoresis
Abstract
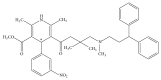
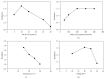
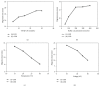
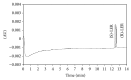
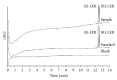
An enantioselective method based on capillary electrophoresis (CE) using cyclodextrin (CD) as chiral selector was developed and validated for determination of lercanidipine (LER) enantiomers, a drug calcium channel blocker which exerts antihypertensive effects of long duration, in a pharmaceutical formulation. Optimum separation of LER enantiomers was obtained on a 50 cm × 50 μm id capillary using a sodium acetate buffer solution 200 mmol/L pH 4.0 containing 10 mmol/L of 2,3,6-o-methyl-β-cyclodextrin (TM-β-CD) as background electrolyte. The capillary temperature and voltage were 15°C and 25 kV, respectively, hydrodynamic injection and detection at 237 nm. Linearity was obtained in the range 12.5-100 μg/mL for both enantiomers (r ≥ 0.995). The RSD (%) and relative errors (E, %) obtained in precision and accuracy studies (intraday and interday) were lower than 5%. After validation, the method was applied to quantify the enantiomers of LER in commercial tablets and the results were satisfactory in terms of accuracy and precision, both less than 5%. Therefore, this method was found to be appropriate for enantioselective quality control of LER enantiomers in pharmaceutical formulations.
Figures
References
-
- Notarbartolo A., Rengo F., Scafidi V., Acanfora D. Long-term effects of lercanidipine on the lipoprotein and apolipoprotein profile of patients with mild-to-moderate essential hypertension. Current Therapeutic Research: Clinical and Experimental. 1999;60(4):228–236. doi: 10.1016/s0011-393x(00)88518-5. - DOI
-
- Godfraind T. Classification of calcium antagonists. The American Journal of Cardiology. 1987;59(3):11B–23B. - PubMed
-
- Goodman G. A., Rall T. W., Tweenies A. S., Taylor P. The Pharmacological Basis of Therapeutics. 9th. New York, NY, USA: McGraw-Hill:; 1992.
-
- Buckley N., Dawson A., Whyte I. Calcium channel blockers. Medicine. 2007;35(11):599–602. doi: 10.1016/j.mpmed.2007.08.025. - DOI
LinkOut - more resources
Full Text Sources
Other Literature Sources

