Parametrial fat tissue from high fat diet-treated SKH-1 mice stimulates transformation of mouse epidermal JB6 cells
- PMID: 25821644
- PMCID: PMC4376014
- DOI: 10.4172/2157-2518.1000183
Parametrial fat tissue from high fat diet-treated SKH-1 mice stimulates transformation of mouse epidermal JB6 cells
Abstract
Our previous studies indicated that decreasing visceral adipose tissue by surgical removal of the parametrial fat pads inhibited UVB-induced carcinogenesis in SKH-1 mice fed a high fat diet (HFD), but not a low fat diet (LFD) indicating that the parametrial fat tissue from mice fed a HFD played a role in skin carcinogenesis.
Objective: In the present study, we sought to investigate how a HFD may influence the intrinsic properties of the parametrial fat tissue to influence UVB-induced skin tumor formation.
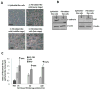
Methods and results: Immunohistochemical staining, adipokine array, and flow cytometry showed that parametrial fat tissue from mice fed a HFD had a higher density of macrophage-fused dead adipocytes (crown-like structures), more adipokines, and stimulated the production of more reactive oxygen species compared with parametrial fat tissue from mice fed a LFD. These differences between parametrial fat tissue from mice fed a HFD and LFD were associated with their effect on the in vitro transformation of mouse epidermal JB6 cells. Our results indicated that fat tissue filtrate (an aqueous filtrate made from the parametrial fat pad) from mice fed a HFD enhanced the conversion of JB6 cells from an epithelial-like morphology to cells with a fibroblast-like morphology to a greater extent than fat tissue filtrate from mice fed a LFD. Studies indicated that the fibroblast-like cells had decreased levels of E-cadherin, increased levels of Twist as assayed by western blot. Fat tissue filtrate made from the parametrial fat tissue of mice fed a HFD had 160% more transforming activity than that from mice fed a LFD and formed malignant mesenchymal tumors in vivo.
Conclusion: These studies provide the first in vitro demonstration of a parametrial fat tissue-induced transformation of an epidermal cell.
Keywords: JB6 cell transformation; high fat diet; parametrial fat tissue.
Figures


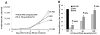

Similar articles
-
Surgical removal of the parametrial fat pads stimulates apoptosis and inhibits UVB-induced carcinogenesis in mice fed a high-fat diet.Proc Natl Acad Sci U S A. 2012 Jun 5;109(23):9065-70. doi: 10.1073/pnas.1205810109. Epub 2012 May 21. Proc Natl Acad Sci U S A. 2012. PMID: 22615388 Free PMC article.
-
The effects on weight loss and gene expression in adipose and hepatic tissues of very-low carbohydrate and low-fat isoenergetic diets in diet-induced obese mice.Nutr Metab (Lond). 2016 Nov 8;13:78. doi: 10.1186/s12986-016-0139-1. eCollection 2016. Nutr Metab (Lond). 2016. PMID: 27826354 Free PMC article.
-
Carvacrol prevents diet-induced obesity by modulating gene expressions involved in adipogenesis and inflammation in mice fed with high-fat diet.J Nutr Biochem. 2012 Feb;23(2):192-201. doi: 10.1016/j.jnutbio.2010.11.016. Epub 2011 Mar 29. J Nutr Biochem. 2012. PMID: 21447440
-
Dietary supplementation with blueberry partially restores T-cell-mediated function in high-fat-diet-induced obese mice.Br J Nutr. 2018 Jun;119(12):1393-1399. doi: 10.1017/S0007114518001034. Br J Nutr. 2018. PMID: 29845904
-
High fat diet consumption differentially affects adipose tissue inflammation and adipocyte size in obesity-prone and obesity-resistant rats.Int J Obes (Lond). 2018 Mar;42(3):535-541. doi: 10.1038/ijo.2017.280. Epub 2017 Nov 20. Int J Obes (Lond). 2018. PMID: 29151595 Free PMC article.
Cited by
-
The Adipocyte-Macrophage Relationship in Cancer: A Potential Target for Antioxidant Therapy.Antioxidants (Basel). 2023 Jan 4;12(1):126. doi: 10.3390/antiox12010126. Antioxidants (Basel). 2023. PMID: 36670988 Free PMC article. Review.
-
Fibroblast growth factor receptor is a mechanistic link between visceral adiposity and cancer.Oncogene. 2017 Nov 30;36(48):6668-6679. doi: 10.1038/onc.2017.278. Epub 2017 Aug 7. Oncogene. 2017. PMID: 28783178 Free PMC article.
-
The Relationship between Leptin, the Leptin Receptor and FGFR1 in Primary Human Breast Tumors.Cells. 2020 Oct 1;9(10):2224. doi: 10.3390/cells9102224. Cells. 2020. PMID: 33019728 Free PMC article.
References
-
- Siegel R, Ward E, Brawley O, Jemal A. Cancer statistics, 2011: the impact of eliminating socioeconomic and racial disparities on premature cancer deaths. CA Cancer J Clin. 2011;61:212–36. - PubMed
-
- Ibiebele TI, van der Pols JC, Hughes MC, Marks GC, Green AC. Dietary fat intake and risk of skin cancer: a prospective study in Australian adults. Int J Cancer. 2009;125:1678–84. - PubMed
-
- Ibiebele TI, van der Pols JC, Hughes MC, Marks GC, Williams GM, Green AC. Dietary pattern in association with squamous cell carcinoma of the skin: a prospective study. Am J Clin Nutr. 2007;85:1401–8. - PubMed
-
- Black HS. Influence of dietary factors on actinically-induced skin cancer. Mutat Res. 1998;422:185–90. - PubMed
-
- Black HS, Lenger W, Phelps AW, Thornby JI. Influence of dietary lipid upon ultraviolet-light carcinogenesis. Nutr Cancer. 1983;5:59–68. - PubMed
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
