Oxyntomodulin regulates resetting of the liver circadian clock by food
- PMID: 25821984
- PMCID: PMC4426666
- DOI: 10.7554/eLife.06253
Oxyntomodulin regulates resetting of the liver circadian clock by food
Abstract
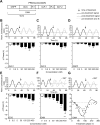
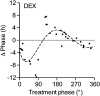
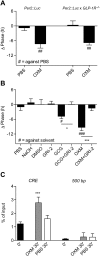
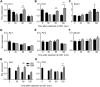
Circadian clocks coordinate 24-hr rhythms of behavior and physiology. In mammals, a master clock residing in the suprachiasmatic nucleus (SCN) is reset by the light-dark cycle, while timed food intake is a potent synchronizer of peripheral clocks such as the liver. Alterations in food intake rhythms can uncouple peripheral clocks from the SCN, resulting in internal desynchrony, which promotes obesity and metabolic disorders. Pancreas-derived hormones such as insulin and glucagon have been implicated in signaling mealtime to peripheral clocks. In this study, we identify a novel, more direct pathway of food-driven liver clock resetting involving oxyntomodulin (OXM). In mice, food intake stimulates OXM secretion from the gut, which resets liver transcription rhythms via induction of the core clock genes Per1 and 2. Inhibition of OXM signaling blocks food-mediated resetting of hepatocyte clocks. These data reveal a direct link between gastric filling with food and circadian rhythm phasing in metabolic tissues.
Keywords: Per; biochemistry; cell biology; circadian clock; clock genes; food resetting; liver; mouse.
Conflict of interest statement
The authors declare that no competing interests exist.
Figures


Comment in
-
Feeding time.Elife. 2015 May 27;4:e08166. doi: 10.7554/eLife.08166. Elife. 2015. PMID: 26012377 Free PMC article.
References
-
- Akhtar RA, Reddy AB, Maywood ES, Clayton JD, King VM, Smith AG, Gant TW, Hastings MH, Kyriacou CP. Circadian cycling of the mouse liver transcriptome, as revealed by cDNA microarray, is driven by the suprachiasmatic nucleus. Current Biology. 2002;12:540–550. doi: 10.1016/S0960-9822(02)00759-5. - DOI - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Molecular Biology Databases

