Novel role of mitochondrial manganese superoxide dismutase in STAT3 dependent pluripotency of mouse embryonic stem cells
- PMID: 25822711
- PMCID: PMC5380158
- DOI: 10.1038/srep09516
Novel role of mitochondrial manganese superoxide dismutase in STAT3 dependent pluripotency of mouse embryonic stem cells
Abstract
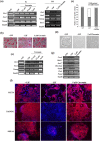
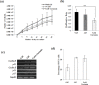
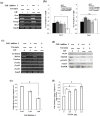
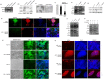
Leukemia Inhibitory Factor (LIF)/Signal transducer and activator of transcription 3 (STAT3) signaling pathway maintains the stemness and pluripotency of mouse embryonic stem cells (mESCs). Detailed knowledge on key intermediates in this pathway as well as any parallel pathways is largely missing. We initiated our study by investigating the effect of small molecule Curcumin on various signalling pathways essential for self-renewal. Curcumin sustained the LIF independent self-renewal of mESCs and induced pluripotent stem cells (miPSCs) in a STAT3 activity dependent manner. Gene expression analysis showed LIF/STAT3 and redox signaling components to be majorly modulated. Amongst ROS genes, expression of Manganese Superoxide Dismutase (MnSOD) specifically relied on STAT3 signaling as evidenced by STAT3 inhibition and reporter assay. The silencing of MnSOD, but not Cu-ZnSOD expression, resulted in the loss of mESC pluripotency in presence of LIF, and the overexpression of MnSOD is sufficient for maintaining the expression of pluripotent genes in the absence of STAT3 signaling. Finally, we demonstrate MnSOD to stabilize the turnover of pluripotent proteins at the post-translational level by modulating proteasomal activity. In conclusion, our findings unravel a novel role of STAT3 mediated MnSOD in the self-renewal of mESCs.
Conflict of interest statement
The authors declare no competing financial interests.
Figures



Similar articles
-
Foxm1 mediates LIF/Stat3-dependent self-renewal in mouse embryonic stem cells and is essential for the generation of induced pluripotent stem cells.PLoS One. 2014 Apr 17;9(4):e92304. doi: 10.1371/journal.pone.0092304. eCollection 2014. PLoS One. 2014. Retraction in: PLoS One. 2019 Jul 5;14(7):e0219580. doi: 10.1371/journal.pone.0219580. PMID: 24743237 Free PMC article. Retracted.
-
Leukemia inhibitory factor-induced Stat3 signaling suppresses fibroblast growth factor 1-induced Erk1/2 activation to inhibit the downstream differentiation in mouse embryonic stem cells.Stem Cells Dev. 2013 Apr 15;22(8):1190-7. doi: 10.1089/scd.2012.0229. Epub 2013 Jan 30. Stem Cells Dev. 2013. PMID: 23205673
-
Gbx2, a LIF/Stat3 target, promotes reprogramming to and retention of the pluripotent ground state.J Cell Sci. 2013 Mar 1;126(Pt 5):1093-8. doi: 10.1242/jcs.118273. Epub 2013 Jan 23. J Cell Sci. 2013. PMID: 23345404
-
Regulation of embryonic stem cell self-renewal and pluripotency by leukaemia inhibitory factor.Biochem J. 2011 Aug 15;438(1):11-23. doi: 10.1042/BJ20102152. Biochem J. 2011. PMID: 21793804 Free PMC article. Review.
-
Cytokine signalling in embryonic stem cells.APMIS. 2005 Nov-Dec;113(11-12):756-72. doi: 10.1111/j.1600-0463.2005.apm_391.x. APMIS. 2005. PMID: 16480448 Review.
Cited by
-
Manganese Superoxide Dismutase Gene Expression Is Induced by Nanog and Oct4, Essential Pluripotent Stem Cells' Transcription Factors.PLoS One. 2015 Dec 7;10(12):e0144336. doi: 10.1371/journal.pone.0144336. eCollection 2015. PLoS One. 2015. PMID: 26642061 Free PMC article.
-
LIF-activated Jak signaling determines Esrrb expression during late-stage reprogramming.Biol Open. 2018 Jan 22;7(1):bio029264. doi: 10.1242/bio.029264. Biol Open. 2018. PMID: 29212799 Free PMC article.
-
Human Trefoil Factor 3 induces the transcription of its own promoter through STAT3.Sci Rep. 2016 Jul 25;6:30421. doi: 10.1038/srep30421. Sci Rep. 2016. PMID: 27453253 Free PMC article.
-
Mitochondrial Superoxide Dismutase Specifies Early Neural Commitment by Modulating Mitochondrial Dynamics.iScience. 2020 Sep 15;23(10):101564. doi: 10.1016/j.isci.2020.101564. eCollection 2020 Oct 23. iScience. 2020. PMID: 33083732 Free PMC article.
-
Rational fusion design inspired by cell-penetrating peptide: SS31/S-14 G Humanin hybrid peptide with amplified multimodal efficacy and bio-permeability for the treatment of Alzheimer's disease.Asian J Pharm Sci. 2024 Aug;19(4):100938. doi: 10.1016/j.ajps.2024.100938. Epub 2024 Jun 24. Asian J Pharm Sci. 2024. PMID: 39253611 Free PMC article.
References
-
- Evans M. J. & Kaufman M. H. Establishment in culture of pluripotential cells from mouse embryos. Nature. 292, 154–156 (1981). - PubMed
-
- Thomson J. A. et al. Embryonic stem cell lines derived from human blastocysts. Science. 282, 1145–1147 (1998). - PubMed
-
- Mitsui K. et al. The homeoprotein Nanog is required for maintenance of pluripotency in mouse epiblast and ES cells. Cell. 113, 631–642 (2003). - PubMed
-
- Nichols J. et al. Formation of pluripotent stem cells in the mammalian embryo depends on the POU transcription factor Oct4. Cell. 95, 379–391(1998). - PubMed
-
- Chen X. et al. Integration of external signaling pathways with the core transcriptional network in embryonic stem cells. Cell. 133, 1106–1117 (2008). - PubMed
Publication types
LinkOut - more resources
Full Text Sources
Other Literature Sources
Miscellaneous

