Mitochondrial ATAD3A combines with GRP78 to regulate the WASF3 metastasis-promoting protein
- PMID: 25823022
- PMCID: PMC4828935
- DOI: 10.1038/onc.2015.86
Mitochondrial ATAD3A combines with GRP78 to regulate the WASF3 metastasis-promoting protein
Abstract
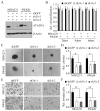
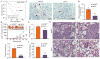
AAA domain containing 3A (ATAD3A) is an integral mitochondrial membrane protein with unknown function, although we now show that high-level expression is associated with poor survival in breast cancer patients. Using a mass spectrometry approach we have demonstrated that ATAD3A interacts with the WASF3 metastasis-promoting protein. Knockdown of ATAD3A leads to decreased WASF3 protein levels in breast and colon cancer cells. Silencing ATAD3A also results in loss of both cell anchorage-independent growth and invasion and suppression of tumor growth and metastasis in vivo using immuno-compromised mice. HSP70 is responsible for stabilizing WASF3 in the cytoplasm, but inactivation of HSP70 does not lead to the loss of WASF3 stability at the mitochondrial membrane, where presumably it is protected through its interaction with ATAD3A. In response to endoplasmic reticulum (ER) stress, increases in the GRP78 protein level leads to increased WASF3 protein levels. We also show that ATAD3A was present in a WASF3-GRP78 complex, and suppression of GRP78 led to destabilization of WASF3 at the mitochondrial membrane, which was ATAD3A dependent. Furthermore, ATAD3A-mediated suppression of CDH1/E-cadherin occurs through its regulation of GRP78-mediated WASF3 stability. Proteolysis experiments using isolated mitochondria demonstrates the presence of the N-terminal end of WASF3 within the mitochondria, which is the interaction site with the N-terminal end of ATAD3A. It appears, therefore, that stabilization of WASF3 function occurs through its interaction with ATAD3A and GRP78, which may provide a bridge between the ER and mitochondria, allowing communication between the two organelles. These findings also suggest that pharmacologic inhibition of ATAD3A could be an effective therapeutic strategy to treat human cancer.
Conflict of interest statement
Figures
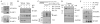



References
-
- Sossey-Alaoui K, Su G, Malaj E, Roe B, Cowell JK. WAVE3, an actin-polymerization gene, is truncated and inactivated as a result of a constitutional t(1;13)(q21;q12) chromosome translocation in a patient with ganglioneuroblastoma. Oncogene. 2002;21:5967–5974. - PubMed
-
- Rotty JD, Wu C, Bear JE. New insights into the regulation and cellular functions of the ARP2/3 complex. Nat Rev Mol Cell Biol. 2013;14:7–12. - PubMed
-
- Sossey-Alaoui K, Head K, Nowak N, Cowell JK. Genomic organization and expression profile of the human and mouse WAVE gene family. Mamm Genome. 2003;14:314–322. - PubMed
-
- Bisi S, Disanza A, Malinverno C, Frittoli E, Palamidessi A, Scita G. Membrane and actin dynamics interplay at lamellipodia leading edge. Curr Opin Cell Biol. 2013;25:565–573. - PubMed
-
- Sossey-Alaoui K, Li X, Ranalli TA, Cowell JK. WAVE3-mediated cell migration and lamellipodia formation are regulated downstream of phosphatidylinositol 3-kinase. J Biol Chem. 2005;280:21748–21755. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Molecular Biology Databases
Miscellaneous

