Phosphorylation events in the multiple gene regulator of group A Streptococcus significantly influence global gene expression and virulence
- PMID: 25824840
- PMCID: PMC4432744
- DOI: 10.1128/IAI.03023-14
Phosphorylation events in the multiple gene regulator of group A Streptococcus significantly influence global gene expression and virulence
Abstract
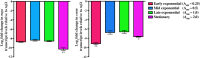
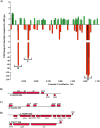
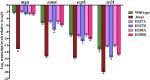
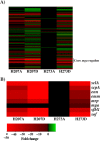
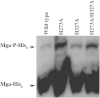
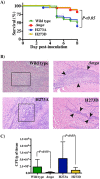
Whole-genome sequencing analysis of ∼800 strains of group A Streptococcus (GAS) found that the gene encoding the multiple virulence gene regulator of GAS (mga) is highly polymorphic in serotype M59 strains but not in strains of other serotypes. To help understand the molecular mechanism of gene regulation by Mga and its contribution to GAS pathogenesis in serotype M59 GAS, we constructed an isogenic mga mutant strain. Transcriptome studies indicated a significant regulatory influence of Mga and altered metabolic capabilities conferred by Mga-regulated genes. We assessed the phosphorylation status of Mga in GAS cell lysates with Phos-tag gels. The results revealed that Mga is phosphorylated at histidines in vivo. Using phosphomimetic and nonphosphomimetic substitutions at conserved phosphoenolpyruvate:carbohydrate phosphotransferase regulation domain (PRD) histidines of Mga, we demonstrated that phosphorylation-mimicking aspartate replacements at H207 and H273 of PRD-1 and at H327 of PRD-2 are inhibitory to Mga-dependent gene expression. Conversely, non-phosphorylation-mimicking alanine substitutions at H273 and H327 relieved inhibition, and the mutant strains exhibited a wild-type phenotype. The opposing regulatory profiles observed for phosphorylation- and non-phosphorylation-mimicking substitutions at H273 extended to global gene regulation by Mga. Consistent with these observations, the H273D mutant strain attenuated GAS virulence, whereas the H273A strain exhibited a wild-type virulence phenotype in a mouse model of necrotizing fasciitis. Together, our results demonstrate phosphoregulation of Mga and its direct link to virulence in M59 GAS strains. These data also lay a foundation toward understanding how naturally occurring gain-of-function variations in mga, such as H201R, may confer an advantage to the pathogen and contribute to M59 GAS pathogenesis.
Copyright © 2015, American Society for Microbiology. All Rights Reserved.
Figures





References
Publication types
MeSH terms
Substances
Associated data
- Actions
Grants and funding
LinkOut - more resources
Full Text Sources
Molecular Biology Databases
Miscellaneous

