Expression of xCT and activity of system xc(-) are regulated by NRF2 in human breast cancer cells in response to oxidative stress
- PMID: 25827424
- PMCID: PMC4392061
- DOI: 10.1016/j.redox.2015.03.003
Expression of xCT and activity of system xc(-) are regulated by NRF2 in human breast cancer cells in response to oxidative stress
Abstract
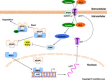
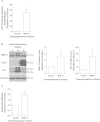
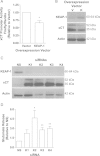
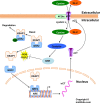
Cancer cells adapt to high levels of oxidative stress in order to survive and proliferate by activating key transcription factors. One such master regulator, the redox sensitive transcription factor NF E2 Related Factor 2 (NRF2), controls the expression of cellular defense genes including those encoding intracellular redox-balancing proteins involved in glutathione (GSH) synthesis. Under basal conditions, Kelch-like ECH-associated protein 1 (KEAP1) targets NRF2 for ubiquitination. In response to oxidative stress, NRF2 dissociates from KEAP1, entering the nucleus and binding to the antioxidant response element (ARE) in the promoter of its target genes. Elevated reactive oxygen species (ROS) production may deplete GSH levels within cancer cells. System xc(-), an antiporter that exports glutamate while importing cystine to be converted into cysteine for GSH synthesis, is upregulated in cancer cells in response to oxidative stress. Here, we provided evidence that the expression of xCT, the light chain subunit of system xc(-), is regulated by NRF2 in representative human breast cancer cells. Hydrogen peroxide (H2O2) treatment increased nuclear translocation of NRF2, also increasing levels of xCT mRNA and protein and extracellular glutamate release. Overexpression of NRF2 up-regulated the activity of the xCT promoter, which contains a proximal ARE. In contrast, overexpression of KEAP1 repressed promoter activity and decreased xCT protein levels, while siRNA knockdown of KEAP1 up-regulated xCT protein levels and transporter activity. These results demonstrate the importance of the KEAP1/NRF2 pathway in balancing oxidative stress in breast cancer cells through system xc(-). We have previously shown that xCT is upregulated in various cancer cell lines under oxidative stress. In the current investigation, we focused on MCF-7 cells as a model for mechanistic studies.
Keywords: Hydrogen peroxide; KEAP1; NRF2; Oxidative stress; System x(c)(−); xCT.
Copyright © 2015 The Authors. Published by Elsevier B.V. All rights reserved.
Figures

Similar articles
-
Sigma 1 receptor regulates the oxidative stress response in primary retinal Müller glial cells via NRF2 signaling and system xc(-), the Na(+)-independent glutamate-cystine exchanger.Free Radic Biol Med. 2015 Sep;86:25-36. doi: 10.1016/j.freeradbiomed.2015.04.009. Epub 2015 Apr 25. Free Radic Biol Med. 2015. PMID: 25920363 Free PMC article.
-
Signal transducer and activator of transcription 3 and 5 regulate system Xc- and redox balance in human breast cancer cells.Mol Cell Biochem. 2015 Jul;405(1-2):205-21. doi: 10.1007/s11010-015-2412-4. Epub 2015 Apr 21. Mol Cell Biochem. 2015. PMID: 25896132
-
Transcription factor Nrf2-mediated antioxidant defense system in the development of diabetic retinopathy.Invest Ophthalmol Vis Sci. 2013 Jun 6;54(6):3941-8. doi: 10.1167/iovs.13-11598. Invest Ophthalmol Vis Sci. 2013. PMID: 23633659 Free PMC article.
-
Cystine-glutamate antiporter xCT as a therapeutic target for cancer.Cell Biochem Funct. 2021 Mar;39(2):174-179. doi: 10.1002/cbf.3581. Epub 2020 Aug 4. Cell Biochem Funct. 2021. PMID: 32749001 Review.
-
The role of Nrf2 in oxidative stress-induced endothelial injuries.J Endocrinol. 2015 Jun;225(3):R83-99. doi: 10.1530/JOE-14-0662. Epub 2015 Apr 27. J Endocrinol. 2015. PMID: 25918130 Review.
Cited by
-
Transcriptome profiles of latently- and reactivated HIV-1 infected primary CD4+ T cells: A pooled data-analysis.Front Immunol. 2022 Aug 26;13:915805. doi: 10.3389/fimmu.2022.915805. eCollection 2022. Front Immunol. 2022. PMID: 36090997 Free PMC article.
-
[The role of ferroptosis in chronic diseases].Zhejiang Da Xue Xue Bao Yi Xue Ban. 2020 May 25;49(1):44-57. doi: 10.3785/j.issn.1008-9292.2020.02.24. Zhejiang Da Xue Xue Bao Yi Xue Ban. 2020. PMID: 32621416 Free PMC article. Review. Chinese.
-
GPX4 restricts ferroptosis of NKp46+ILC3s to control intestinal inflammation.Cell Death Dis. 2024 Sep 19;15(9):687. doi: 10.1038/s41419-024-07060-3. Cell Death Dis. 2024. PMID: 39300068 Free PMC article.
-
Mechanism of ferroptosis in breast cancer and research progress of natural compounds regulating ferroptosis.J Cell Mol Med. 2024 Jan;28(1):e18044. doi: 10.1111/jcmm.18044. Epub 2023 Dec 22. J Cell Mol Med. 2024. PMID: 38140764 Free PMC article. Review.
-
Molecular pathology underlying the robustness of cancer stem cells.Regen Ther. 2021 Mar 25;17:38-50. doi: 10.1016/j.reth.2021.02.002. eCollection 2021 Jun. Regen Ther. 2021. PMID: 33869685 Free PMC article. Review.
References
-
- Balendiran G.K., Dabur R., Fraser D. The role of glutathione in cancer. Cell Biochemistry and Function. 2004;22:343–352. - PubMed
-
- Bannai S. Induction of cystine and glutamate transport activity in human fibroblasts by diethyl maleate and other electrophilic agents. Journal of Biological Chemistry. 1984;259:2435–2440. - PubMed
-
- Sasaki H., Sato H., Kuriyama-Matsumura K., Sato K., Maebara K., Wang H., Tamba M., Itoh K., Yamamoto M., Bannai S. Electrophile response element-mediated induction of the cystine/glutamate exchange transporter gene expression. Journal of Biological Chemistry. 2002;277:44765–44771. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials

